
Medtronic pays $84M in damages for Edwards TAVI patent infringement

The Medical Device Business Journal — Medical Device News & Articles | MassDevice
 Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
Welcome to MassDevice's Cardiovascular Hub, your resource for the latest news in cardiac implants, structural heart, heart valves, stents and more. Stay informed about the innovations and business news shaping the cardiovascular device industry.
🫀 Narrow your focus with these categories:
Cardiac Implants | Structural Heart | Pulsed-Field Ablation
Here are top cardiology features from MassDevice and its sister publications (and scroll down to read the latest cardiology device news from MassDevice):
+ LVADs save lives: So why aren’t more available?
+ How Medtronic uses nitinol to improve the structure and effectiveness of heart devices
+ How Abbott designed the world’s first dual-chamber leadless pacemaker system
+ J&J used RWE for expanded indications — and you can, too
+ 7 cardiology startups you need to know

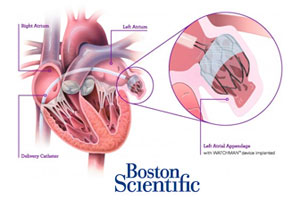
The Watchman heart implant made by Boston Scientific (NYSE:BSX) improved quality-of-life measures more than warfarin in atrial fibrillation patients after 1 year, according to a study in the Journal of the American College of Cardiology.

Medical device giant Medtronic (NYSE:MDT) won European regulatory approval for its Engager transcatheter aortic valve implantation system, the company announced today.
The CE Mark indication includes approval for transapical delivery of the valve in treatment of patients with aortic stenosis who are too sick to undergo traditional surgery.
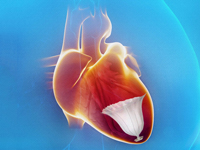
CardioKinetix said it’s raised $48 million in its Series E round with the closure of a 2nd tranche worth $23 million and plans to use the proceeds to further its Parachute ventricular partitioning device, which is in a pivotal trial.

Medtronic (NYSE:MDT) said the FDA approved an early trial of its native outflow tract transcatheter pulmonary valve under a new agency program for early feasibility studies.
The TPV device is designed for patients born without a right ventricle-to-pulmonary artery conduit, which provides blood to the lungs, as an alternative to open heart surgery.

German medical device maker Biotronik won European regulatory approval for its Ilesto 7 line of MRI-friendly heart implants.
The Ilesto 7 line includes cardiac resynchronization therapy defibrillator and one of the world’s smallest implantable cardioverter-defibrillators, according to Biotronik.

OrbusNeich is taking a recent patent infringement win against Boston Scientific (NYSE:BSX) even further, filing lawsuits in both Germany and the Netherlands to allege patent infringement over stent technology.

