 Advanced Cooling Therapy said today it won expanded CE Mark approval in the European Union for its Esophageal Cooling Device, now cleared to be used for up to 120 hours in patients needing warming or cooling.
Advanced Cooling Therapy said today it won expanded CE Mark approval in the European Union for its Esophageal Cooling Device, now cleared to be used for up to 120 hours in patients needing warming or cooling.
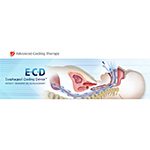
The ECD is designed to modulate and control patient temperature through a triple lumen system inserted into the esophagus, with 2 lumens attached to temperature modulation equipment and a 3rd lumen to provide gastric decompression and drainage.
“We are encountering more and more clinicians who want to use our ECD for longer durations of treatment, for cooling, warming, or maintaining patients at ideal body temperature when clinically necessary. Being able to use the ECD for up to 120 hours will allow clinicians to better manage patient temperature in a variety of circumstances and enhance patient care,” Clinical services director Maria Gray said in a press release.
The expansion boosts time of use for the ECD from 36 hours, the company said.
“We are using the ECD for an increasing number of patient conditions that require longer treatment, and look forward to being able to optimize patient care with this expanded indication,” Dr. Ahmed Hegazy of the London Health Sciences Centre said in prepared remarks.
In January, Advanced Cooling Therapies said it won FDA 510(k) clearance for its ECD-02 esophageal cooling device, the company’s 2nd-gen version of the ECD.
The Chicago-based company said the device is designed to be “rapidly inserted” by trained healthcare professionals, similar to standard gastric tubes. ACT added that no other product on the market are approved for use in the esophagus for temperature modulation.
Last June, Advanced Cooling Therapy won de novo clearance from the FDA for its ECD-01, claiming it as the sole temperature modulation device to use the esophagus.
The ECD won CE Mark approval in the European Union in 2014 and is on the market in Canada and Australia.

