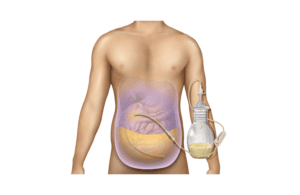
 BD (NYSE:BDX) today said it has received FDA 510(k) clearance for its PeritX peritoneal catheter system.
BD (NYSE:BDX) today said it has received FDA 510(k) clearance for its PeritX peritoneal catheter system.
Franklin Lakes, N.J.-based BD designed the catheter system to drain symptomatic, recurrent non-malignant ascites, which causes a build-up of fluid in the abdomen.
Get the full story on our sister site, Medical Tubing + Extrusion.

