Bioventus said it acquired the OsteoAMP product line from Advanced Biologics for an undisclosed amount.
OsteoAMP is a tissue-based product used to promote natural bone growth and healing in the spine, according to a press release.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
Bioventus said it acquired the OsteoAMP product line from Advanced Biologics for an undisclosed amount.
OsteoAMP is a tissue-based product used to promote natural bone growth and healing in the spine, according to a press release.

Once-scorned biomaterials maker FzioMed is getting another day with the FDA to discuss its Oxiplex spinal surgery gel, which was rejected by U.S. healthcare officials in 2008.
FzioMed will meet June 10, 2014, with the FDA’s Medical Device Dispute Resolution Panel to discuss the "approvability" of the Oxiplex gel, which has been on the European market since 2012 and is approved for sale in 70 countries. The product has already been used in more than 350,000 surgeries, FzioMed said.

Wright Medical (NSDQ:WMGI) said it’s not optimistic about an FDA dispute resolution panel on its Augment biologic bone graft product, now slated for May, and would quickly move to cut costs at the BioMimetic Therapeutics subsidiary it bought back in 2012.

Mazor Robotics (NSDQ:MZOR) announced a deal with an unnamed group purchasing organization for its flagship Renaissance robotic surgery system.

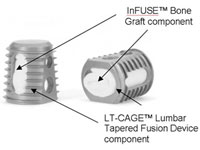

Federal pre-emption statutes have thus far stalled personal injury complaints over Medtronic’s (NYSE:MDT) troubled Infuse bone-growth protein, but new lawsuits may find their way to trial by claiming that the medtech titan is liable for injuries because it engaged in off-label marketing.

