EnteroMedics (NSDQ:ETRM) is looking to control metabolic disease by attacking the problem closer to its source – human behavior.
"We tried to understand why people became over-weight and obese, and what was the underlying mechanism," CEO Mark Knudson told MassDevice.com. "As we looked into it more closely, we found that the autonomic nervous system – which is the body’s house-keeping nervous system, the nervous system that works outside of a person’s volitional control … was internally involved in the control of hunger, feelings of fullness, control of glucose, control of blood pressure."
Using a new twist on neuromodulation therapies, which stimulate certain nerves to activate a response, the company’s Maestro implantable therapy aims to block certain physiological mechanisms to help patients be less hungry and feel full after consuming less food.
Using proprietary VBloc, or vagal blocking therapy, EnteroMedics’ Maestro device stimulates the Vagus nerve, located between the stomach and the esophagus, in order to alter a patient’s eating habits by modifying their physiological cravings.
"We actually treat a patient’s problem, which is the hunger and the feeling of fullness, so that they can immediately start changing their relationship to food and make a lifelong change while not having their whole lifestyle interrupted," Knudson said.
In an exclusive interview with MassDevice.com, Knudson and EnteroMedics CFO Greg Lea told us about the device, the company’s expansion goals for the "Metabolic Disease Crescent" and the bid for U.S. regulatory approval.
MassDevice: Tell me a little bit about EnteroMedics’ history and how you developed Vagal blocking therapy.
Mark Knudson: EnteroMedics was started as part of an incubator group called the Venturi Group in St. Paul, Minnesota. We started out as a focus on trying to develop a therapy for one of the largest and fastest growing problems facing the health care universe – not just in the U.S., but in the world – and that is obesity, diabetes and hypertension, often referred to as metabolic disease.
"We actually treat a patient’s problem which is the hunger and the feeling of fullness, so that they can immediately start changing their relationship to food and make a lifelong change while not having their whole lifestyle interrupted."
The current therapies for obesity, which is in many ways the underlying cause of much of the Type II diabetes and hypertension in modern society, are very patient-unfriendly and difficult for patients to live with. As we’ve seen, the number of patients who are either under-weight or obese just continues to grow worldwide.
So we tried to understand why people became over-weight and obese, and what was the underlying mechanism for the transformation of this problem of overweight into the development of diabetes, the lack of being able to control of blood sugar or blood pressure problems. And as we looked into it more closely, we found that the autonomic nervous system – which is the body’s house-keeping nervous system, the nervous system that works outside of a person’s volitional control, the system that keeps your digestive system functioning, keeps your lungs functioning, the heart functioning, all of things that belong below the level of consciousness, that’s called the autonomic nervous system – was internally involved in the control of not just these things that I talked about but it was internally involved in the control of hunger, feelings of fullness, control of glucose, control of blood pressure.
And what you realize is that, we felt that that was certainly a target of big interest to us in terms of developing an effective therapy that will actually address the problems people have with controlling their weight and controlling what’s going on, which is they are either hungry all the time or they never feel full. We targeted, therefore, the vagus nerve, because the vagus nerve becomes more and more active, the more hungry you are. And when you eat the eat the vagus nerve is at its most active in that it is controlling how your stomach expands, it’s controlling the secretion of the digestive enzymes, all the way from the acid in your stomach to the pancreatic enzymes that digest the food as it continues through the digestive tract. It is very, very active.
So we realized that what we needed to do was find a way to down-regulate that nerve. So we began looking at the other side of neural modulation, which is called neural-blocking. And we’re the first company that’s developed really effective, commercially viable and available neural-blocking technology for the treatment of any problem. And because of that we have very strong intellectual property in the area of treating obesity and metabolic disease as well as several other gastrointestinal disorders. And from this we are going to launch what we call the Maestro system which delivers VBloc therapy. VBloc therapy uses very high frequency electrical signals to block the naturally occurring signals on the vagus nerve, on the autonomic nerve, from the brain to the stomach and the rest of the gut and from the gut and stomach to the brain.
We do this intermittently all through the day – it’s on 5 minutes, off 5 minutes and then it’s off during the sleeping hours of the patient. We do this for 2 reasons: one is so that the patients don’t have side effects. If we completely turn off that nerve then the whole system does not work, the patient has side effects. And if we keep it turned off 24 hours a day, then the body figures a way to work around the fact that it doesn’t have the vagus anymore and most of the effect on reducing hunger and early fullness and other metabolic effects falls away.
We began work on the this – we filed our 1st patents in the fall of the 2003, did our 1st implant in 2006, and are now commercial outside the U.S. We have a CE Mark and we’re approved in Australia and we are in the last 6 months before breaking the blind on the U.S. physical trial. Assuming that that comes out well, we will be submitting a PMA application in 2013. That is a quick overview of the company and how we got where we are.
"We think of it as a 1st-in-class, 2nd-generation neuroscience-based approach to the treatment of the biggest problem facing the modern society."
MassDevice: What have you seen Vagal blocking therapy do for patients as far as outcomes?
Knudson: Patients tend to lose weight in a controlled and calculable manner. It controls their hunger and it makes them feel full on much less food. Some of the other therapies that are out there, some of them surgical therapies, are punitive. In other words they close up most of the stomach and the lower part of the intestines, so people cannot eat very much before they get clogged up or even have a lot of very unpleasant side effects. Many of them are still very hungry for the most part and so it is hard for them to change their relationship to food. We actually treat a patient’s problem which is the hunger and the feeling of fullness, so that they can immediately start changing their relationship to food and make up a lifelong change while not having their whole lifestyle interrupted.
They can still eat, they can still go out and have a steak or have a piece of bread, which they cannot do with, for example, the adjustable gastric band. They can still have a glass of wine with their dinner, which they can’t with some of the other procedures. They can still do this they just don’t need as much and they are not as hungry all the time – they do not snack between meals. As a result of this they lose between 30-40% of excess their weight by 1 year and if they happen to be diabetic their blood glucose is under control almost immediately and if they are hypertensive, they have a very good chance that their hypertension will be under control almost immediately as well.
So it is very patient-friendly, life enhancing, lifestyle-enhancing therapy. We think of it as a 1st-in-class, 2nd-generation neuroscience-based approach to the treatment of the biggest problem facing the modern society.
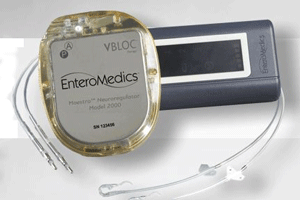
MassDevice: Now, I had seen the Maestro system described as a pacemaker-like device. Describe the device and tell me about the implantation procedure.
Knudson: It is a pacemaker-like device. There is a neural-regulator with leaves attached to the neural-regulator implanted in a minimally-invasive, laparoscopic, outpatient procedure It’s performed by the same surgeons today that do either a banding procedure or a surgical procedure, and it is very patient-friendly. It can be turned off if it needs to be and the patients tolerate it extremely well. We’ve had a very strong safety track record with no serious adverse events in over 700 patients now around the world.
And so it is extremely safe and the patients really like it. It is implanted under the skin. The neural regulator is implanted under the skin at the rib cage and then the leads are attached to it laparoscopically and implanted and around the Vagus nerve right above the stomach. It’s a very simple procedure, the surgeons are required to go through a very short training. They use the same tools and techniques that they would use for other procedures, and so we anticipate that it will become widely supported by the bariatric community once we get approval here in U.S.
We are commercialized outside the U.S., we just recently announced our 1st agreements with 2 distributors – 1 in the Middle East and 1 in Australia – and they are recruiting patients. We’ve had our 1st commercial implants of patients that occurred in the Middle East. While we do not have a lot of experience in the commercial environment, we do have a lot experience in the clinical setting.
MassDevice: What is the patient experience like? Can they feel it when it goes off?
Greg Lea: There are a few patients. Right after the system is activated in a few of the patients they say they can feel a little tingling that tends to go away over time, and some patients tolerate it very well. Most of the patients tell us that, should they not have the system activated or not have the system function properly they can tell then, because their old habits are back. But for the majority of patients, they get to the point where they do not feel anything or report anything with the device being active.
MassDevice: Is there some sort of ceiling to this therapy? Will it work for as long as it is implanted and active?
Lea: Yeah actually that is why we do the 5 minutes on and 5 minutes off, and off at night. Every morning when the patient wakes up it’s like receiving new therapy, the body feels like it’s receiving new therapy so every 24 hours, it’s like new therapy. So we’re seeing patients who want to continue this therapy indefinitely. They view this as their way to support their lifestyle and not go back to the old habits, and as I mentioned before, we have patients now hitting year 7 with this technology implanted.
The blocking signal is a very high frequency signal, that as Mark referred to earlier, you know, just disrupts or blocks the natural occurring signals of the Vagus nerve to the brain from the stomach and from the brain to the stomach.

MassDevice: The device is rechargeable as well, so is there a point at that which it would need replacement, outside of any maintenance issues or if something happened?
Knudson: Yes, we believe somewhere between 5, 7, 10 years the patient would have to go through a very standard, small procedure of a small incision replacing the neural regulator battery. And it is rechargeable, our experience is that most patients will recharge their device like they do their cell phone – once a day or once every other day. And recharges in 30-45 minutes and it’s a very simple charge. They lay a transmit coil on the skin where the device is implanted and, through radio frequency, the charge is delivered. Again it takes 30-45 minutes to do that.
MassDevice: And how long does a charge last?
Lea: The charge will last you know, roughly a week or more. If it went without charging for that period time, they also have a portable battery pack that is actually the charging device, for the neural regulator. So they probably have a week and a half to 2 weeks off a charge, between the battery pack and the device itself, before they have to get to a charging station, which is a very typical charging station that you set the battery pack in and charges it quickly.
MassDevice: You had mentioned that EnteroMedics is the 1st to develop this. Is anybody following in your footsteps?
Knudson: Actually you know there was a conference about 2 weeks ago, in Salt Lake City about high frequency blocking, and there are several companies that are looking to develop neural blocking therapies for other applications. So there are several companies that are looking to do this as well as several academic institutions, for example, Case Western University.
MassDevice: I understand you have got CE mark, you have got approval on Australia, are you working toward approval in any other market as well?
Knudson: I mentioned that we are working towards U.S. approval, but our main focus right now is on what we call the Metabolic Disease Crescent, which is from Australia up to Indo-China, China, India into the Middle East. Because of the genetic makeup of that population, they tend to develop Type II diabetes at much lower BMIs than do European populations. So we’re an ideal device there because metabolic surgery as it is practiced right now in the U.S. before the approval of our device, really the only choices available to you are anatomy-altering surgeries where you cut out a perfectly healthy stomach or you re-route the entire intestinal tract or you put in an adjustable gastric band that you have to have adjusted 4 to 7 times a year, and you have to wait to lose some amount of weight before you have effectiveness.
Our device serves to address that problem immediately for those patients, and it gives them the full flexibility of a device that’s neuroscience-based that’s ‘s fully programmable. For example, if you are a young woman and you are pre-diabetic, your diabetes is under control and you want to have children, you just turn the device off and then once you had a child, you can turn it back on to help lose weight and move forward with your life. So, it gives patients all the flexibility they could ever want without permanently altering their anatomy or their nutritional status. By that I mean the ability to absorb critical nutrients, like protein and micro nutrients like vitamins and minerals.
MassDevice: So where exactly is the Maestro system commercially available now?
Knudson: It is commercially available in Australia and the Gulf coast countries, in the Middle East, and there are a couple select, key thought leaders in Europe who are also using the device. Although we do not have an active marketing program in Europe.
MassDevice: Tell me about the pivotal trial in the U.S., the Recharge trial.
Knudson: The trial is a randomized and controlled trial, a multi-center trial. We have 233 subjects enrolled. It is randomized 2:1 to treatment and control and the control arm is a sham surgery with a sham device implanted. The last patient was implanted at the end of last year and we expect to break the blind at the end of this year, and make an announcement in early 2013 with the results of that trial and assuming positive results. We expect to move forward then with PMA application to the FDA in the 1st half of 2013.
The Recharge trial is our 2nd pivotal trial. We did a previous pivotal trial called the Empowered trial with the 1st generation device. Which had a various positive outcomes for safety, but it missed the comparative efficacy endpoint because we’re the 1st company that’s ever had to do a randomized-control, placebo-control trial and because of the design of that trial and because of the way neural devices are both arms of the trial actually had some energy delivered because of the safety checks and the heat checks required.
It turns out that it resulted in an unanticipated therapeutic effect in the control arm. Actually both arms of the trial pretty much met our statistical targets for end points. Therefore comparative endpoint was not met but it really gave us what we needed to design the 2nd trial. And that’s why the 2nd trial is a sham control trial. But those patients are continuing and are doing very well losing weight and we are looking forward to breaking deadline to our 2nd generation device and making it widely available in the U.S.
MassDevice: EnteroMedics hit some big milestones this year with 1st commercial sales, and the 1st commercial implants. Can you tell me a little more about these recent wins?
Lea: Well, it was according to our schedule. We received the final approval of all the modules, all components of our technology in Australia in January and we had already signed the distribution agreement. And so, our first commercial sales went in to the distributor in Australia. We followed that very quickly with signing the distribution agreement in the Middle East and commercialized the products there, and they moved on a very rapid schedule and they had the 1st commercial implant shortly thereafter. We expect to have continuing commercial sales in both those geographies as we go through the year.
We will announce those commercial sales as part of our quarterly financials and we will continue, as we have in the past, to announce when it is relevant and important as we did around OUS, feasibility trials, and most notably we are still conducting a trial outside the U.S. in diabetics and we will continue announcing data as that becomes available. Our next big milestone will be with the announcement of the positive data from a U.S. pivotal trial at the beginning of next year.
MassDevice: Are there any other big expectations for the rest of this year and the next few years?
"[The medical device tax] has a huge impact on us and our hiring plans and our launch plans in the U.S. We will do a lot more outside the U.S. before we can even think about a doing something in the U.S. just because of the impact of that tax."
Lea: Obviously, the next year will be a very big year for us. Once we announce the results of the pivotal trial, we will continue to announce the schedule we are on with FDA, including our PMA submission, and we will move towards panel with anticipation that if we continue to expect positive results, which we are, then we will have that submission complete and have panel review late 2013, and hopefully approval by the FDA and in the first half of 2014.
We will continue to announce expansion outside the U.S. We would expect to see expansion in the other geographies as our resources permit into Europe and potentially more of the Middle East. India would be very important area for us to look at with the resources we have, as we move into 2013. And so that’s really what we are focused on for the next year and half.
MassDevice: I know that EnteroMedics doesn’t have U.S. sales on the horizon just yet but is the impending medical device tax that is contained in the Affordable Care Act on your radar?
Knudson: Absolutely, especially for a start-up company like ourselves. It’s a tax on sales, so it directly impacts the cash we can use, the people we can hire. We definitely cannot use as much as we otherwise could because it comes right off the top even when we are not profitable, even we don’t have the cash to do everything we want to do. So, it has a huge impact on us and our hiring plans and our launch plans in the U.S. We will do a lot more outside the U.S. before we can even think about a doing something in the U.S. just because of the impact of that tax.
MassDevice: Are there any other measures aside from doing more outside the U.S. before you get to the U.S.? Are there any other measures you are taking to prepare?
Knudson: I don’t think so. One of our plans is to this year – we hope, just depends on whether we have the resources to get to it, if not then early next – to expand our CE mark to including independent indications for diabetes and hypertension in order to expand our reach outside the U.S. We will also do that in Australia and then continue to work with thought leaders around the world as they try to bring new and expanding technology to their patients.

