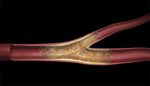
Tryton Medical said today that its Side Branch Stent designed to treat coronary bifurcation lesions has been used in its 1st commercial procedure, performed by Dr. Martin Leon and Dr. Ajay Kirtane at the New York-Presbyterian Hospital .
The side-branch stent is designed to be compatible with any conventional drug-eluting stent in the main vessel, and comes in sizes ranging from 2.5mm to 3.5mm, the Durham, N.C.-based company said.
“Dr. Leon and the cath lab team at Columbia University Medical Center played an integral role investigating the Tryton Side Branch Stent through extensive clinical research leading to the recent FDA approval. We are honored to have the team at Columbia perform the first U.S. commercial case and appreciate their guidance during the Tryton Clinical Trials and leadership they provide for the interventional community around the world. Tryton looks forward to working with Cordis to ensure interventional cardiologists in the U.S. have access to a dedicated bifurcation device,” CEO Shawn McCarthy said in a press release.
Earlier this month, Tryton Medical won pre-market approval from the FDA for its its side-branch stent for treating coronary bifurcation lesions and inked an exclusive U.S. distribution deal with Cardinal Health (NYSE:CAH) subsidiary Cordis.
Although the company’s device failed to meet its primary endpoint for target vessel failure in a pivotal study with a target vessel failure rate of 17.4%, a subsequent analysis of patients with lesions suitable for a 2.5mm stent showed an 11.3% TVF rate, compared with 15.6% for the control cohort treating with provisional stenting.

