
When Novocure won FDA approval for its Tumor Targeting Field cancer therapy system, the company introduced the oncology world to an entire new modality for treatment.
Cancer patients today are most famliar with surgery, radiation and chemotherapy as thearpy optoins, but certain patients can also opt for Novocure’s non-invasive electromagnetic therapy, which promises to stop cancer cells in their tracks without adding a new suite of side effects.
The TTF system, currently only FDA-approved for certain deadly brain cancers, includes a series of white bandages that are applied to the head and connected to a wearable battery-pack via a series of wires.
The device generates continuous electromagnetic fields that are tuned to the frequency of the specfic cancer, unleashing a silent attack that prevents the targeted cells from dividing without affecting the healthy cells nearby. And it does this without significantly affecting mental function, causing pain or preventing the patient from going to work. The most common side effect amounts to little more than a skin rash.
The TTF device won FDA approval in April 2011 to treat glioblastoma brain tumors, the most common and aggressive form of primary brain tumor in the U.S., after an FDA panel determined that the system improved quality of life for cancer patients
In this in-depth interview with MassDevice.com, Novocure executive chairman Bill Doyle explained how the therapy works, what it’s like introducing physicians to an entirely new platform and the company’s bid for expanded indication to target non-small cell lung cancer.
MassDevice: What is your background?
William Doyle: I am the executive chairman of Novocure. I have been involved with the company since 2003. I’m also the managing director of a medical device focused venture capital firm called WSP Ventures. And we are the largest investors in Novocure as well.
MassDevice: That’s how you originally became involved with Novocure?
WD: Yes, it’s an investment that we made.
I’m an engineer by training and spent probably the most relevant part of my career before WSP Ventures at Johnson & Johnson (NYSE:JNJ). I was responsible for the business development and medical devices group for most of the 90s.
MassDevice: Were you with Novocure from the beginning?
WD: For a long time, but the company was founded by a gentleman named Dr. Yoram Palti, a professor emeritus at Technion in Israel. Palti founded the company based on a theory that he advanced that – or I should say invented is probably a better word than advanced – but a theory that he first proposed that electrical fields – intermediate frequency low-intensity electric fields – could be used to kill dividing cancer cells.
“You know about startups in the garage? He was literally in the basement of his house.”
You know about the startups in the garage? His was literally in the basement of his house. He started doing cell cultures in the basement of his house and he demonstrated the effect in cell culture. And then he founded a company with a few loyal employees to further demonstrate the principle in preclinical animal experiments. I was the first professional investor in the company but we had gotten involved in 2003. We made our first investment in early 2004 and that financed the 1st human pilot trials in the company.
MassDevice: Is the professor still involved with Novocure?
WD: Yes, he is. He is our chief technology officer.
MassDevice: Tell me about a little bit more about Tumor Treating Fields. What are they exactly?
WD: This is a subject of which many PhDs could be written.
Essentially, when cells divide they undergo mitosis. They round up and they form little balls. The nuclear membrane disintegrates and the nuclear material, the chromosomes, line up in the middle of this rounded-up cell. It’s in time that a structure called the microtubule spindle begins to form in the cytoplasm and the microtubule spindle is literally the cellular structure that attaches to the chromosomes and pulls them apart, pulls the genetic material in half and pulls half of it into one daughter cell and half of it into the other daughter cell. This is a physical process.
The microtubule spindles are formed from protein tubule subunits that are among the most highly charged in the body, so they’re highly charged dipoles. What Palti first hypothesized, and has been proven again in the dish and then in animal models and then in human, is that when an electric field is applied – and the electric field has to be applied at just the right frequency for the right amount of time in just the right way, so it’s very specific in particular – but when all those conditions are met, those tubular subunits line up, if you will, in the selected fields and are prevented from forming the normal microtubule spindle required for division.
So what happens is the cells end up trying to divide and they try and try and in the experiments as observed – this takes about 4 to 5 hours – but after trying and failing for after about 4 to 5 hours, the cells undergo a programmed cell death. So rather than having 1 cancer cell become 2, 2 become 4, 4 become 8 etc., 1 becomes 0.
And we have demonstrated that over time tumor growth is inhibited and, in fact, tumors can shrink in the presence of TT Fields.
MassDevice: How do you prevent the fields from targeting healthy cells?
WD: TT Fields focus on the microtubules. Some of the most effective chemotherapies have also targeted the microtubule spindle and in particular the [chemotherapy drugs called] taxols.
The issue with taxols historically – which have been a very powerful weapon – because they’re so powerful, the side effect profile has been difficult with these drugs. The toxicity has been difficult.
Tumor Treating Fields avoid that in 2 ways. First of all it’s a regional therapy, so the fields are applied using external transducer rays. They look like bandages with wires coming from them that surround the region of the tumor. So in case of brain cancer – which is the cancer from which we have our 1st FDA approval – our patients wear a transducer ray on each side of their head and then the back of their head in order to expose the brain to the TT Field.
The 1st reason for the extremely less toxicity is that the fields are only applied in the region. So you wouldn’t expect to have any effect, let’s say, on the bone marrow or the GI tract if you’re only treating the brain.
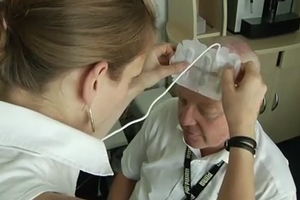
Most importantly is the 2nd issue, which is that the cancer cells affected, or the divided cells affected, are affected at the specific frequency of the field. And that frequency is inversely related to cell size, meaning the smaller the cell the higher the frequency. And generally speaking cancer cells are smaller than normal healthy cells. So the frequencies required to kill, in this case, [glioblastomas] are approximately 4 times greater than the frequency that you would use, let’s say, if you wanted to affect healthy lining of the GI tract.
GBM, the brain cancer, has a different frequency than lung cancer which has a different frequency than breast cancer. All of which have a different frequency than the typical healthy tissues that one would be concerned about.
And so the net of all this has been in all of our clinical trial work we don’t see any local, regional or systemic toxicity. The 1 device-related adverse event that we do observe is a slight contact dermatitis under the transducer ray. So basically you’ve got a little redness under the bandage.
And we saw that in about 16% of the patients in our phase 3 trial. Again, when you think about side effects from cancer therapies, a little redness under the bandages is considered to be mild.
MassDevice: How exactly does a patient use this? Does a patient borrow this from a hospital? Do they wear it all day?
WD: So basically when this is on, it’s killing cancer cells. When it’s off it’s not. And because of the low toxicity, patients can essentially have chronic long term therapy for their cancer rather than the episodes of radiation therapy or the doses of chemotherapy that are, by nature, episodic.
So the therapy is delivered as a box with a battery. The 2 together weigh about 5½ pounds today. That is our 2nd generation, which is smaller than our 1st, and like all things electronic it will be smaller and lighter in the next generation that’s currently designed. Each one of the transducer rays, as I described in the brain cancer, the full transducer rays has a wire that comes from the bandage, and those wires plug into the box.
The patient carries the box in either a backpack or over the shoulder. It has a battery, or if they are at home or at work they can plug it in. So it’s very much like carrying your laptop around, is a way to think about it. When you have it in your briefcase or your purse, it’s battery powered. And when you’re stationary, you plug it in. The patients are encouraged to use the device as much as possible.
There is no sensation, there is no tingling or pain or burning or anything like that. They just have to wear the bandages and they have to use the transducer rays and they have to make sure that the batteries are changed and that it’s plugged in. And the therapy is delivered over a long period of time.
MassDevice: Is it possible to wear it too much or is it literally as much as you can, you should?
WD: As much as you can, you should. Now patients who take breaks, they take a break to shower, they take breaks to exercise, we – and this is based on our preclinical work – suggest that they should aim for 20 hours a day of therapy.
MassDevice: And this is continuous for the as long as they still have evidence of cancer?
WD: So this is one question that we don’t have the perfect answer for, which is when to stop. With the particularly deadly cancers – the brain cancer – the patients are advised to use the device as long as they can if they can physically tolerate the device. In our current newly diagnosed Phase III trial it’s designed for up to 2 years of therapy.
MassDevice: And what happens after that?
WD: What we don’t know today is whether 23 months is enough or 24 or whether 25 is better than 24. What we’ve seen in our Phase II research is that after about 2 years patients who have had good responses, those responses are long-term durable even after they stop the therapy. But as I said we just are not in a position to say exactly what the perfect time is. It’s a decision that the patient and his or her doctor make as they approach the 2-year mark.
MassDevice: So when you say that the responses were durable and long-lasting, does that mean that they continued to be cancer-free even when not using the device?
WD: Correct.
MassDevice: About how long afterwards was that shown for?
WD: So data have been published, but in a small single arm Phase II trial. This was 10 patients in Europe. The patients received – and this is very similar to our Phase III protocol-type – the patients in that particular Phase 2 received therapy for up to 18 months. And the responses at 5 years in the trial had still not reached the median. So there were several years, those were 3 years beyond the time of stopping the therapy at least.
MassDevice: That’s pretty impressive.
WD: Again, this was Phase II. I want to make sure I appropriately describe it. So it’s Phase II, not randomized, but the results were extremely promising. And it’s based on those promising results, that’s why we made the enthusiastic decision to go forward with a randomized Phase III trial to truly prove it.
MassDevice: I know you guys won FDA premarket approval last year, and you mentioned it was just for glioblastoma. Is it specific to end-stage patients or patients who have exhausted other options?
WD: Yeah, so let me make sure I’m very clear about this. Our first Phase III trial, the one that’s completed and the one for which we received FDA approval – that trial which was our first Phase III, was for recurrent GBM patients. So these were patients offered enrollment after they had failed standard therapy.
And standard therapy in this disease includes surgery for those patients that are eligible, then high-dose hovering radiation with a drug called Temozolomide, or Temodar the brand name. And then after they finish the radiation in Temodar they receive maintenance Temodar, so they receive Temodar alone.
Virtually every patient with this disease, it does come back or recur. The median time for recurrence is about 7 months. So this really is one of the worst cancers. And then at that point in time the recurrence, then they’re eligible for Tumor Treating Field therapy. So those are the recurring patients.
Now in the trial that we just published, we divided patients into 2 groups, the 1st group received TT Field therapy alone. So it was a mono-therapy with no other therapy. And then the 2nd group received the – it was not a placebo control it was an active control – they received the best, it’s called “best physician choice.” There’s a small group of chemotherapies that are given as a 2nd-line therapy and depending on the patient and the setting, the physician makes that choice.
So that was the comparison in the 1st trial. And that is the approval that we have received. So the approval is for patients who have recurred after receiving standard therapy and it is for using Novocure as a stand-alone mono-therapy.
The 2nd trial – the trial that is underway and the trial that we started based on the Phase II results that I just described – is for newly diagnosed patients. So these are patients when they’re 1st diagnosed with the diseases. And then in the newly diagnosed patients, they still receive surgery if they are eligible, they still receive radiation and Temodar during radiation. And during the maintenance phase of Temodar we add Novocure. So it’s Novocure plus chemo. And in the trial, that’s compared to chemo alone.
And that’s the trial that’s underway and we expect to finish recruiting that trial toward the end of next year.
And this is typical when you have a brand new modality like Tumor Treating Fields, we’re typically required to start with the sickest patients first. And we’re typically required to start with a therapy alone as a mono-therapy. And there’s some logic to that in that in this way there is no confusion about what is resulting in the effect.
However, given that the therapy has essentially no toxicity and we found in both preclinical – so in vitro and in vivo preclinical animal work – an advantage to using TT Fields with chemotherapy, the degrees to which they work together depends on the specific chemo, but they work together.
So it makes great sense that the 2nd or the next trial is to move upstream to where the patients are healthier and where we can use the therapy together with their chemo. And that’s what we’re currently undertaking in this second Phase III trial.
MassDevice: So right now it seems all your research is being conducted on brain cancer specifically. Are you looking at other cancers as well?
WD: The first cancers as we just discussed is GBM, the deadly brain cancer. We have done an additional Phase II trial in patients with non-small cell lung cancer. So this was a trial that was a multicenter trial done in Switzerland with 42 patients.
In this trial we treated with Tumor Treating Fields and the chemotherapy. And we also showed very promising results so the patients – it was a Phase II trial, 42 patients, not randomized – but compared to historical data of the chemo alone, the results were extremely promising. And so Novocure is currently planning a Phase III trial in non-small cell lung cancer.
We’ve also done a tremendous amount of laboratory work over the last 10 years. And have shown that in the laboratory setting, Novocure is effective against a broad range of cancers. We’ve done a pilot trial in breast cancer where we have seen very encouraging results. But right now the company is planning and making decisions about the next trials. But non-small lung cancer is certainly going to be one that we’re focused on.
We’re going to be focused on not only primary brain cancer GBM but brain metastases. So these are cancers that have spread often from lung cancer, breast cancer or melanoma. We are looking at the other difficult-to-treat solid tumors of the trunk, so ovarian cancer for instance; pancreatic cancer is another one where we have shown effectiveness in the laboratory in vitro.
And so right now we are planning our next series of clinical trials. And what we imagine is over the next few years is that we will be doing Phase III trials in these additional cancers in order to determine if we’re effective in randomized trial.
MassDevice: Is there any type of cancer that this couldn’t potentially extend to?
WD: So every cancer line that we have used applied TT Fields to in the laboratory we’ve shown effectiveness – meaning that we have killed the cancer cells. Now in real-world application, we need, as we described in the beginning and you’ll see if you look at some of the films, we need to surround the region of the cancer with the transducer rays in order to get the fields there. Practically, it’s not going to be a therapy for circulating tumors, so tumors in the blood or lymph nodes.
It’s also not going to be a therapy for cancers of the bone marrow. Now one of the good things about this is that the long bones – as opposed to the flat bones of the skull – the long bones are a natural shield for TT Fields. The bad news is we are not likely to use the therapy to treat cancers of the bone marrow. The good news is it has no effect on the blood cell-producing tissues of the bone marrows.
We don’t see those side effects that you often see, with the fatigue and the increased infection rates as blood cells are affected. What we’re focused on are the cancers of the head, neck and trunk where we can get transducer rays in place.
MassDevice: Do you have any estimates on when you might start pursuing some of other cancers?
WD: We’re working on those things right now. As you can imagine from some of the announcements that we’ve made, we’re busy – extremely busy – with the launch of the recurrent GBM product and the ongoing Phase III and newly diagnosed GBM. We’re actively planning the lung cancer trial and the brain mets trials. But I don’t think we’re at a point where we can specifically set target dates. We’re just not that far along in the planning.
MassDevice: I did see the press release on the addition of new centers of excellence. It was 8 of them, right?
WD: Yeah, yeah so we had 7 in the first wave and then 8 in the second wave.
MassDevice: What does that mean for the company as far as a milestone?
WD: I think it’s an incredible milestone. We are bringing a brand new modality. So it’s different than a new drug, which is a great accomplishment as well. But a great drug – it’s another bottle with a pill in it or it’s another vile. The infrastructure exists, the community understands how to administer those products. We’re bringing something completely new.
So we need to train the centers. We need to learn what the best training methodology is. We need to establish the support infrastructure for the centers and for the patients. And it’s all going extremely well. But we’re determined to go in the highest possible way. And we’d rather go slow to ensure that we get it right.
And so that is why you see there is very deliberate phased, what we call “leadership launch.” Starting with 7 centers and now adding the next 8. I think if you view the list you can see these are among the top centers in the US. I mean we are very, very pleased to be partnering with this group. We will continue to add centers throughout the rest of this year and next. So this is not the end, it’s the beginning. But we are going to do it very carefully and very deliberately to make sure that every center is supported and every patient is supported.
MassDevice: The current trials bring TT Fields to the frontline in combination with chemo. Is there a future possibility that it could be a frontline standalone?
WD: Particularly with a deadly cancer like GBM, given that we see that 1 plus 1 equals 3, if you will,the 2 – chemotherapy plus TT Fields – are more effective together than either one is separately and TT Fields results in no added toxicity. This will be all established on a cancer by cancer basis. Not only the likely combination, but specifically what is the right chemo? But we think that that is the most likely use that we will see as the trials progress.
MassDevice: So it’s not going to replace chemo, it’s just going to enhance or support it?
WD: Absolutely, absolutely. This is not about replacing chemotherapy, this is about the 2 of them together being more effective than either separately.
MassDevice: Does Novocure have any outside-U.S. clearances?
WD: Yeah, we have CE Mark for recurrent GBM, newly diagnosed GBM and we actually have the CE Mark for non-small cell lung cancer as well. So we received the CE Mark in 2009 for the GBM. And I believe it was 2010 for the non-small cell lung cancer.
MassDevice: Is there anyone else developing these TTF systems?
WD: We are the pioneers in this field. We own an extensive patent portfolio. There are researchers who are working to understanding it better. There is a group at Harvard Medical School, Eric Wong’s group that has published some research in this area. And we know the group at the Karolinksa Institute in Sweden that’s researching this. And we’re starting to see here and there references to other academic groups that have begun to work on this. But we are the only company that is doing this. And, as I said, we are literally the pioneers that are working to bring this to oncologists and patients.
MassDevice: You mentioned earlier that the system is brand new, a completely different type of therapy. Are physicians initially skeptical?
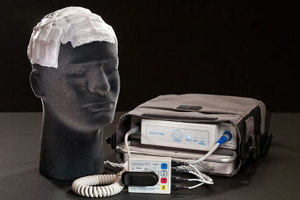
WD: The device itself is a completely new concept but it’s relatively simple.
The typical certification of centers is about a 4-hour process. It’s basically half a day training the staff at the particular cancer center, and that includes training in the basic science, training in the use of the device, as well as taking the appropriate credentialing exams. Then we certify the center.
I think everything new starts off with a skeptical response. I can say that when I 1st learned about this, I was skeptical. I think so was every one of our board members, our employees and every one of our key opinion leaders who’s been involved in this. But that’s just the nature of something new.
We’re very pleased to describe that when people – and this is whether new prospective employees, patients, clinicians, centers – when they sit down, when they study the mechanism of action and understand the basis for it and then look at the data, skeptics become less skeptical and less skeptical and then they become believers.
So I think that’s a process that everyone has to go through. And they go through it with Novocure; I think they go through it probably with iPhones and the next iPad too – you know, “Do I really need that?” But it’s certainly a process that we see.
I think you can see from the centers that now have been trained and they’re prescribing the therapy that this therapy is beginning to find its place as a standard therapy in the tool chest. And I think that this is something that is going to continue. We see the momentum accelerating.
And things like, of course, getting an FDA approval, a published paper in the peer reviewed research, all of these things are part and parcel of establishing the evidence, quite frankly. I wouldn’t expect anybody to believe anything without the evidence. And as a company, that’s what we are working to provide. We have provided quite a bit and we are continuing to provide more, which is why we continue to run Phase III clinical trials.
MassDevice: What’s the reimbursement status for TT Field therapy?
WD: Because it’s brand new there are no existing codes, if you will. So we are going through the same process that any new technology like the new like the new percutaneous valves for instance or the new stents, which I was involved with at J&J, anything new has to go through a process to establish its reimbursement. We are in the midst of that process right now. That process typically starts in the early days but individual applications meaning that on a patient by patient basis the doctors and the patients apply for reimbursement. And then over time as those patterns are established then companies like ours will apply for broader codes. That’s typically a process that takes about 18 months.
But given our efficacy and safety profile and how we compare to the other therapeutic options, we fully expect to establish reimbursement for this therapy.

