
Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with Massdevice +3.
3. Cochlear shares nosedive after hearing implant recall
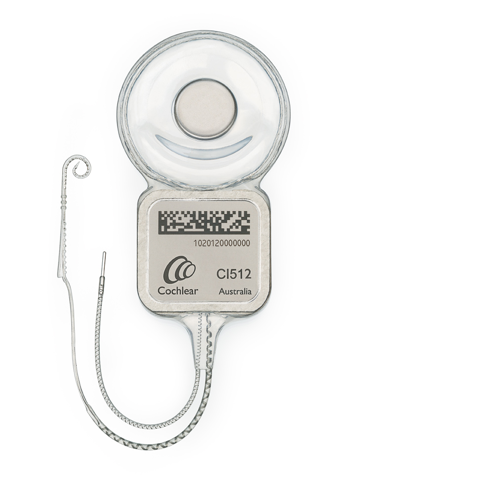
Cochlear Ltd. (ASX:COH) saw a 20 percent sink in its share price when it announced a recall on its Nucleus CI500 series. The voluntary recall was announced after unexplained failure in a number of the Nucleas CI512 implants.
![]()
Sign up to get our free newsletters delivered right to your inbox.
![]()
2. Riata leads fare worse than St. Jude’s leads on
An Irish study finds that wires in St. Jude Medical Inc.’s (NYSE:STJ) Riata defibrillator leads may poke through their insulation cables at rates much higher than reported by company statements.
![]()
1. Mesh makers defend their devices amid FDA probe
Medical device big shot Boston Scientific (NYSE:BSX) urges the FDA to maintain a class II device status on transvaginal mesh despite growing claims that the products do more harm than good.

