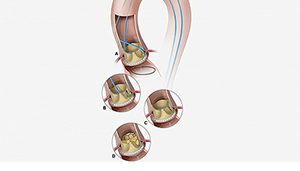
The researchers at NIH’s National, Heart, Lung, and Blood Institute are calling the procedure Bioprosthetic Aortic Scallop Intentional Laceration to prevent Iatrogenic Coronary Artery obstruction (BASILICA). They think the technique could TAVR available to even more high-risk patients who need heart valve procedures.
A leading TAVR company, Edwards Lifesciences, previously projected a $5 billion market by 2021 for the procedure. The advance at NIH could potentially accelerate TAVR’s expansion even more. About 5 million people in the United States each year receive a heart valve disease diagnosis; more than 20,000 of them die, according to American Heart Association statistics related by NIH.
Get the full story on our sister site Medical Design & Outsourcing.

