Updated March 19, 2013, at 11:30 a.m. to reflect that the quote in paragraph 6 came from an ACC press release, not an Edwards press release.

Medical device maker Edwards Lifesciences (NYSE:EW) reported today that its Sapien transcatheter aortic valve implantation system appears durable and effective at 3 years compared with open-heart surgery, but that the risk of valve leakage is "overwhelmingly higher," researchers noted.
The medical device giant today unveiled the latest results from its PARTNER clinical trial, reporting that the Sapien technology proved similar at 3 years to surgical valve replacement for all-cause mortality, symptom improvement and stroke risk.
Procedure-related adverse events, however, remain a concern for the TAVI technology, including valve leakage and short-term stroke rates.
The news didn’t sit well with Wall Street, which sent EW shares down 2.4% to $88.11 as of about 2:30 p.m. EST today.
"In these 1st-generation transcatheter procedures, we have equivalent midterm outcomes between TAVR and the gold-standard surgical valve replacement in high-risk patients with severe aortic stenosis," said Dr. Vinod Thourani, who presented the trial results at today’s American College of Cardiology conference in San Francisco. "However, paravalvular leak continues to increase mortality at 3 years. Physicians should adopt innovative imaging technologies for more accurate sizing to help decrease these leak rates during TAVR."
"Leaks around the valve were common soon after the procedure and were overwhelmingly higher in the TAVR group," Thourani noted in an ACC press release.* "Even mild aortic leakage is associated with a higher mortality rate after any valve replacement procedure."
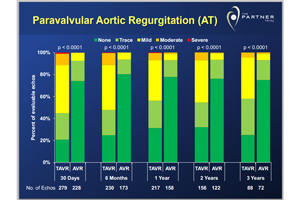
A slide from Edwards Lifesciences’ presentation illustrating the differences in valve leakage between Sapien and surgery.
Click to enlarge.
The results were similar to those released at last year’s ACC conference, when Edwards Lifesciences reported 2-year findings from the PARTNER study.
The new findings highlighted the similarities between TAVI and surgical valve replacement in terms of all-cause mortality, cardiovascular mortality and stroke rates at 3 years.
At 6% the 30-day stroke rates for TAVI were significantly higher than the 3.2% reported for surgery, but TAVI narrowed that gap over the course of time. At 3 years the TAVI stroke rate was 8.2%, compared with surgery at 9.3%.
All-cause mortality was "nearly identical" with TAVI at 44.2% and surgery at 44.8%, and cardiovascular mortality was "statistically indistinguishable" with TAVI at 30.1% and surgery at 30.2%, according to the report.
Phase III of the PARTNER study will follow patients for 5 years, the company noted.

