
Cordis Corp., a Johnson & Johnson (NYSE:JNJ) subsidiary, said it inked an exclusive deal to distribute TriReme Medical’s Chocolate PTA balloon catheter.
The Chocolate PTA device is designed to treat peripheral artery disease
The Medical Device Business Journal — Medical Device News & Articles | MassDevice
By Chris Walker

Cordis Corp., a Johnson & Johnson (NYSE:JNJ) subsidiary, said it inked an exclusive deal to distribute TriReme Medical’s Chocolate PTA balloon catheter.
The Chocolate PTA device is designed to treat peripheral artery disease

California-based TriReme Medical announced this week that the company and its partner Century Medical received Japanese regulatory approval for the GliderXtreme PTA catheter, designed to treat blockages in the arteries.

TriReme Medical landed a regulatory win in Japan with Shonin approval of its Glider PTCA catheter.
TriReme said it plans to begin distribution of the torqueable catheter through its Japanese manufacturing partner, Century Medical.
Pleasanton, Calif.- and Singapore-based TriReme said the Glider device is designed for difficult coronary surgical cases, such as bifurcations and highly stenosed lesions.

The FDA approved a 1st-of-its-kind treatment for Type 2 diabetes, Johnson & Johnson‘s (NYSE:JNJ) Invokana, with the federal watchdog agency citing it as a pioneer in a new class of diabetes drug.

Vascular devices maker TriReme Medical announced a new $18 million financing round in support of the company’s new dilation catheter pipeline and global commercial efforts.
Financing was led by Luminor Capital of Singapore and joined by new Asian investors as well as existing funders, according to a press release.


The University of Michigan is suing St. Jude Medical (NYSE:STJ), accusing the medical device maker of failing to pay $4.3 million in royalties on sales of St. Jude’s Epic and Trifecta cardiovascular valves.
Updated Dec. 23, 2011, to correct that the HeartNavigator is inserted through a small incision in the leg, not in the chest.
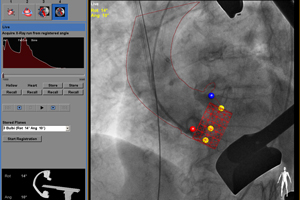 Philips’ HeartNavigator aortic valve replacement system uses x-ray imaging to guide the valve to the heart.
Philips’ HeartNavigator aortic valve replacement system uses x-ray imaging to guide the valve to the heart.Philips Healthcare (NYSE:PHG) landed FDA clearance for its HeartNavigator heart valve replacement system.
Medical City Dallas Hospital became the first U.S. hospital to use the system, which was launched in Europe in the first quarter of this year.

Medtronic Inc. (NYSE:MDT) named GE Healthcare’s former chief, Omar Ishrak, to take over from chairman and CEO William Hawkins, effective June 13.
The news from the Fridley, Minn.-based medical device behemoth came just one day after rival Boston Scientific Corp. (NYSE:BSX) announced that CEO J. Raymond Elliott will be retiring at the end of the year.

