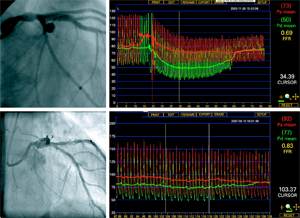
Results from the Fame II study comparing St. Jude Medical‘s (NYSE:STJ) PressureWire fractional flow reserve device with optimal medical care show that FFR-guided coronary interventions are more cost-effective, sending STJ shares up nearly 2% today on Wall Street.