Contego Medical said it pulled in $5.6 million in a Series B round to advance its embolic protection technology for angioplasty balloon and stent-delivery catheters.
Stroke
More good news for stent thrombectomy
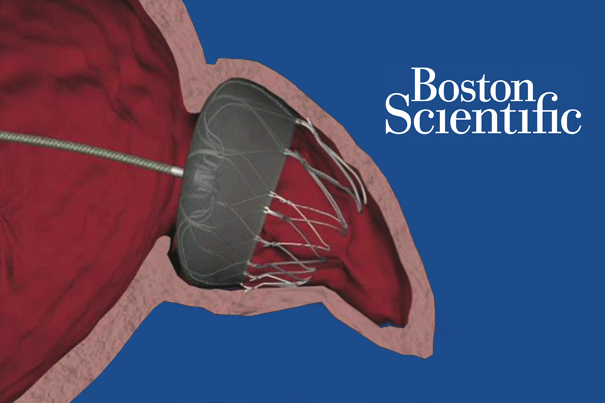
UPDATE: Boston Scientific jumps on Watchman approval
Investors sent shares of Boston Scientific (NYSE:BSX) up nearly 4% today after the medical device company said the FDA approved its long-delayed Watchman anti-stroke device.
Studies: After stroke, clot removal saves brain function
FDA OKs Silk Road Medical’s anti-stroke Enroute device
UPDATED Feb. 10, 2015, with comments from trial investigators and Silk Road Medical.
The FDA today said it granted 510(k) clearance to the anti-stroke Enroute device made by Silk Road Medical.
Mr. Clean: Device makers jump on positive thrombectomy results in stroke trial
Survey: Stroke still a mystery to many
UPDATE: 3rd FDA panel vote barely favors Boston Scientific’s Watchman anti-stroke device
Press Release: REVIVE SE thrombectomy device approved for ischemic stroke patients in China, South Korea and Taiwan

DePuy’s Codman Neuro expands in Asia Pacific with new approvals

Massachusetts-based Codman Neuro is preparing to take Asia Pacific by storm with new regulatory wins in China, South Korea and Taiwan for the company’s Revive SE thrombectomy device.
Study: Afib doubles stroke risk after surgery
Results from a new study suggest that atrial fibrillation (AF) may pose a significant threat to patients undergoing surgery, especially those having non-cardiac procedures.
In an analysis of more than 1.7 million patients, researchers found that perioperative AF was associated with twice the risk of stroke in patients who had non-cardiac surgery. Patients who underwent a cardiac procedure saw a 30% increase in their stroke risk.