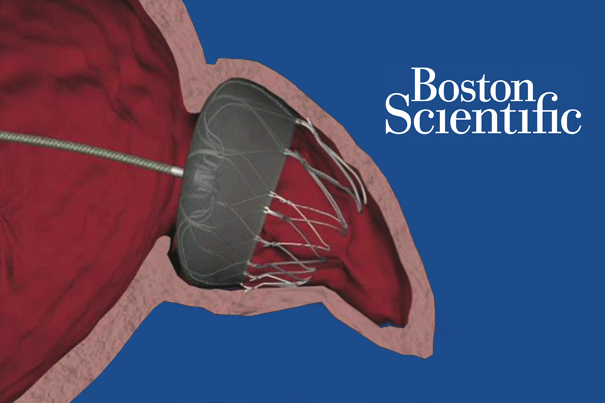
Boston Scientific (NYSE:BSX) shares took a hit yesterday after a Centers for Medicare & Medicaid Services proposal that would limit coverage for the Watchman anti-stroke device. Investors also likely reacted to a pair of Class II recalls from the FDA, sending BSX shares down -4.2% to $18.01 apiece yesterday. Watchman, a transcatheter implant designed to seal off […]
Stroke
Silk Road Medical raises $57 million
Silk Road Medical today said it reeled in a $57 million equity-and-debt round from a group of new and existing backers. Sunnyvale, Calif.-based Silk Road, which makes the Enroute transcarotid artery stent and neuroprotection system, said CRG led the debt round and participated in the equity portion, joined by prior investors Warburg Pincus and The Vertical Group. “The […]
Medtronic launches Arc stroke catheter in the U.S.
Medtronic (NYSE:MDT) said today that it expanded its thrombectomy offering in the U.S. with the Arc catheter, designed for use with its Solitaire stent retriever in treating acute ischemic stroke. Fridley, Minn.-based Medtronic said the Arc catheter won 510(k) clearance from the FDA in July. “The Arc support catheter will complement our Solitaire stent retriever and […]
Penumbra files for $115m IPO
Penumbra, fresh off its latest entry into the peripheral vascular market, last week registered for an initial public offering worth $115 million. Alameda, Calif.-based Penumbra also makes devices for treating neurovascular conditions including hemorrhagic and ischemic stroke. The company reported sales of $125.5 million last year, up 41.3% over 2013. Founded in 2004, Penumbra said it employs […]
FDA warns on SentreHeart’s Lariat anti-stroke device
The FDA today warned healthcare providers about the possibility of potentially deadly adverse effects from the Lariat anti-stroke device made by SentreHeart, saying a review of its adverse events database turned up 45 incidents involving Lariat, including 6 deaths. The Lariat device is designed to close off the heart’s left atrial appendage, to prevent blood clots […]
New Watchman study reports positive data
New data from a study of Boston Scientific (NYSE:BSX)’s Watchman left atrial appendage closure device reports positive results in a study comparing it to warfarin, an anticoagulant drug treatment, in patients with nonvalvular atrial fibrillation. The 2,406-patient study, published in the Journal of the American College of Cardiology, reported a lower rate of hemorrhagic strokes, unexplained and cardiovascular […]
Penumbra wins FDA nod for Ace64 thrombectomy device
Penumbra said today that it has received 510(k) clearance from the FDA for its Ace64 aspiration thrombectomy device, based on data from the Mr. Clean trial and additional randomized controlled stroke trials.