Conventus Orthopaedics, with an FDA clearance in hand, plans a gradual rollout of a new device designed to help repair shoulder fractures in a less invasive way.
Shoulders
FDA clears Tornier’s Simpliciti shoulder repair device
Rotation Medical raises $27M for rotator cuff system

Minnesota-based Rotation Medical plans in coming weeks to advance its commercial efforts for its rotator cuff system thanks to the $27.2 million in raised from new and existing investors.
The oversubscribed Series B financing included backing from new investor Life Sciences Partners who co-led funding with New Enterprise Associations. New investor Pappas Ventures also joined the round among unnamed other backers.
Cayenne Medical nears $6M funding goal

Cayenne Medical is $1 million away from reaching its $6 million fundraising goal, according to a regulatory filing.
Scottsdale, Ariz.-based Cayenne Medical makes medical devices used in knee and shoulder reconstruction surgeries.
Nine unnamed investors ponied up a collective $5 million in the equity and options sale, according to the filing.
Biomet launches U.S. IDE trial for stemless shoulder replacement

Biomet said it’s completed the first U.S. implantation in an investigational device exemption trial of its Comprehensive Nano stemless shoulder device.
The stemless shoulder, which is already on the market in Europe, is based on Biomet’s T.E.S.S. stemless shoulder, which has been on the market since 2004.
FDA says yes to Zimmer’s patient-specific shoulder implant system

LMT Surgical recalls shoulder implants amid 3% fracture rate
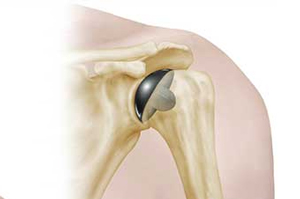
In consultation with Australian health authorities, LMT Surgical issued a warning to all surgeons who implanted the company’s PyroTitan shoulder implant, advising of a reported 3% failure rate in the devices.
Arthrocare gets FDA nod for shoulder suture system | Regulatory Roundup
ArthroCare Corp. (NSDQ:ATRC) landed FDA clearance for its SpeedFix suture system for repairing tears in the shoulder joint, a common injury.
The sutures tout independent bone locking, suture tensioning and suture locking to fasten tissue to bone using double-loaded proprietary MagnumWire sutures.
The company won FDA clearance for their SpeedFix anchor system in November 2010.



