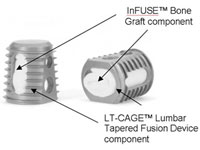
The U.S. Justice Dept. closed the books on a years-long investigation into Medtronic‘s (NYSE:MDT) controversial Infuse bone growth protein, the company announced today.
The office of the U.S. Attorney for the District of Massachusetts and the DoJ abandoned their civil and criminal probe into allegations of off-label marketing and concealed safety risks without finding evidence of wrongdoing, according to a press release.