
Medtronic (NYSE:MDT) could avoid a major headache if the courts continue to allow the doctrine of preemption in product liability lawsuits filed over its InFuse bone-growth protein, according to Fish & Richardson principal José Sierra.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

A California state court ruled out a series of motions seeking to dismiss a lawsuit filed over bone proteins made by Stryker and Medtronic, ruling that federal law does not preempt the case from going forward.


Startup medical device company Tissue Regeneration Systems raised $4.6 million in an equity financing round led by Venture Investors, CEO Jim Fitzsimmons told MassDevice.com.
The round was also supported by the Wisconsin Alumni Research Foundation, the University of Michigan and the state of Wisconsin, Fitzsimmons told us.

Wright Medical (NSDQ:WMGI) shares are down nearly 9% today after the medical device company revealed that the FDA won’t approve its Augment bone graft.
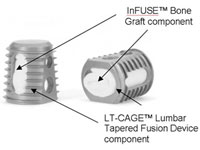

Federal pre-emption statutes have thus far stalled personal injury complaints over Medtronic’s (NYSE:MDT) troubled Infuse bone-growth protein, but new lawsuits may find their way to trial by claiming that the medtech titan is liable for injuries because it engaged in off-label marketing.


