The FDA today warned of potential risks associated with fluid-filled intragastric balloons, produced by ReShape Medical and Apollo Endosurgery, which are used to treat obesity. The federal watchdog said it has received multiple reports of 2 different types of adverse events associated with the balloons, including over-inflation requiring premature removal and the development of acute […]
ReShape Lifesciences
ReShape Medical taps Sirtex prez Mangano for CEO
ReShape Medical taps Sirtex prez Mangano for CEO ReShape Medical said today it tapped former Sirtex president Michael Mangano as its new prez and chief executive officer. During his time at Sirtex, Mangano helped grow the company’s revenue from $34 million to over $130 million. Mangano spoke with MassDevice.com about his time with the company, […]
How Reshape Medical built an alternative to bariatric surgery for obesity | DeviceTalks Podcast
In this episode of DeviceTalks, we speak with Richard Thompson, CEO of ReShape Medical, about the company’s bariatric surgery alternative for people suffering from obesity. In July, San Clemente, Calif.-based ReShape won pre-market approval from the FDA for its dual balloon system, indicated to facilitate weight loss in obese adult patients. The system is designed to be inserted […]
ReShape closes $38m Series D round
ReShape Medical said today it closed a $38 million Series D round of financing, with funds slated to support the commercialization of its ReShape integrated dual balloon weight-loss system. The round was led by HealthCor Partners Management and included participation from Endeavour Vision SA, SV Life Sciences, New Leaf Venture Partners, U.S. Venture Partners and […]
FDA approves ReShape Medical’s weight loss treatment
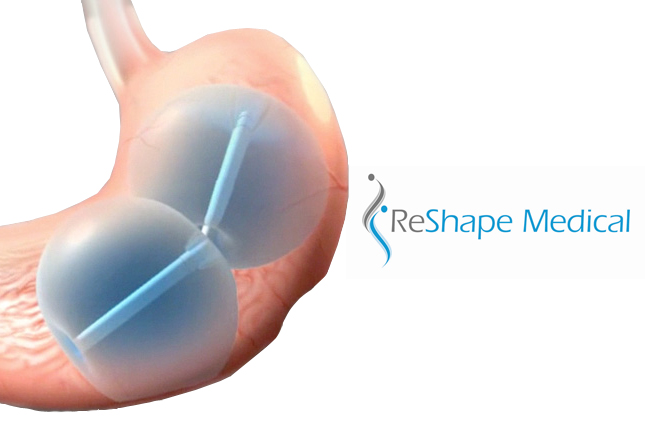
Weight loss company ReShape Medical won FDA premarket approval for its ReShape dual balloon system, indicated to facilitate weight loss in obese adult patients, according to an FDA release. The system is designed to be inserted endoscopically into the stomach, where it remains for 6 months to help patients feel full, San Clemente, Calif.-based ReShape […]
New JenaValve CEO is Boston Scientific vet Carr-Brendel | Personnel Moves
JenaValve said this week that it tapped Victoria Carr-Brendel, former general manager of Boston Scientific‘s (NYSE:BSX) Bayer Interventional business, as its new chief executive officer. Carr-Brendel will replace Jan Keltjens, who served as interim CEO of the transcatheter aortic valve repair-focused company since January 2015. Keltjens will remain with the company as chairman of the board, JenaValve said. […]
EXCLUSIVE: FDA says ReShape Medical can skip advisory panel for obesity balloon device

ReShape Medical, which is developing a balloon system to combat obesity, notched a significant victory in its path to commercialization, CEO Richard Thompson told MassDevice.com today.
Regulators at the FDA waived a requirement that the San Clemente, Calif.-based company appear before an advisory committee for its premarket approval application, Thompson told us. That removes a significant amount of uncertainty from the arduous PMA process for the ReShape Duo device, he said.
Weight loss: ReShape Medical closes $6.75M funding round
ReShape Medical, the medical device maker developing a balloon system to combat obesity has raised nearly $7 million through a debt financing round, according to regulatory filings.
Disinfection robots, Google Glass, artificial pancreas top hospitals’ tech list
Weight loss: ReShape Medical’s Duo balloon system looks to curb obesity
BioVentrix raises $12M for heart attack treatment | MassDevice.com On Call

MASSDEVICE ON CALL — BioVentrix reported a $12.4 million funding round for its Revivent heart failure treatment.
San Ramon, Calif.-based BioVentrix said in an equity filing that the equity round, involving 195 unnamed investors, began Jan. 24 and closed today.