Neuronetics today priced its upcoming initial public offering, expecting to raise approximately $93.5 million to support its NeuroStar transcranial magnetic stimulation system designed to treat patients with major depressive disorder who aren’t responding to medication. The Malvern, Penn.-based company said it is looking to float 5.5 million shares of its common stock at $17 per share […]
Neuronetics
Neuronetics adds up-to $12m over-allotment option to pending IPO
Neuronetics this week added an underwriter’s option to its initial public offering, which could bring in between $10.5 million and $12 million if fully exercised, and gave a rough price range for the offering. The Malvern, Penn.-based company said it is filing for the public offering to expand its commercialization efforts for the NeuroStar transcranial magnetic […]
Neuronetics files for $86m IPO
Neuronetics yesterday registered an initial public offering worth more than $86 million with the U.S. Security & Exchange Commission, saying it plans to expand its commercialization efforts for the NeuroStar transcranial magnetic stimulation device for treating depression. The NeuroStar device uses MRI-strength magnetic fields to stimulate specific areas of the brain associated with mood. The device initially […]
Neuronetics raises $34m for anti-depression device
Neuronetics said it raised $34.3 million in an equity offering slated to help broaden the use of its anti-depression device that relies on magnetic stimulation of the brain.
Neuronetics launches postpartum depression trial

Neuronetics said it’s enrolled 5 patients in an open-label trial of its transcranial magnetic stimulation device for treating postpartum depression.
The Malvern, Pa.-based medical device company makes the NeuroStar TMS device, designed to use a focused magnetic field to stimulate brain cells to treat depression.
Survey: Most companies aren’t ready to implement healthcare reform | MassDevice.com On Call

MASSDEVICE ON CALL — The vast majority of U.S. companies consider rising healthcare costs a "top concern" moving forward, and more than half are under-prepared to implement programs required by new laws.
More than 80% of the companies surveyed named healthcare among the top budgetary concerns, up from the 74% in the last iteration of the study.
Sign up to get our free newsletters delivered straight to your inbox
[sead]
Study: Experimental anorexia treatment with deep brain stimulation may provide lasting results

In what some are calling "a world 1st," researchers reported some success in treating severe anorexia patients via implanted electrodes that delivered electrical energy to the regions of the brain associated with emotion.
The study enrolled 6 patients to receive the experimental treatment, reporting that 3 of the 6 achieved weight gain and 4 of the 6 reported changes in mood, anxiety, control over their urges to binge and purge and other symptoms associated with anorexia.
Neuronetics’ depression therapy device wins CE Mark | Regulatory Roundup
The top 5 stories of the summer, Medtronic responds to the insulin pump hacker, treating depression with electromagnetism | MassDevice.com +3

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with Massdevice +3.
MassDevice Podcast: Neuronetics CEO Bruce Shook
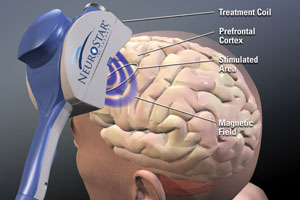
Neuronetics’ NeuroStar TMS therapy system
Neuronetics Inc. is paving the way for a new type of depression therapy, a non-invasive electromagnetic field treatment designed to stimulate brain cells linked to depression.
Bruce Shook, co-founder, president & CEO, talked to MassDevice about pioneering the market for the only FDA-cleared transcranial magnetic stimulation system to date, his company’s NeuroStar TMS system.
The therapy, which won the FDA nod in 2008, is a rarity in the med-tech world: A device-based approach to a psychiatric disorder.



