EDAP TMS SA
FDA panel votes against EDAP’s ultrasound prostate cancer ablation
Press Release: EDAP completes FDA inspection of manufacturing site

LYON, France — EDAP TMS SA (Nasdaq:EDAP), the global leader in therapeutic ultrasound, today announced that the U.S. Food and Drug Administration (FDA) concluded its routine inspection of EDAP’s manufacturing site with no findings nor issuance of Form 483 observations.
EDAP creeps toward FDA approval with clean inspection
MGH researchers gain $30 million grant to build brain implants for treating mental illness | Medtech funding for the week of May 26, 2014
EDAP outlines road to FDA approval for prostate cancer ultrasound therapy

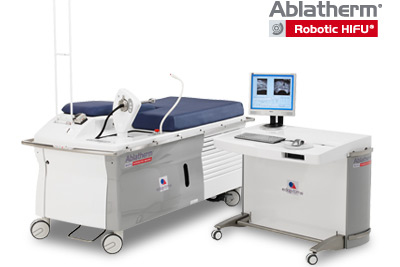
Press Release: EDAP’s Ablatherm-HIFU FDA Panel Meeting Confirmed for July 30, 2014

Prostate cancer: EDAP lands FDA date for therapeutic ultrasound

France-based EDAP TMS SA will get its day with the FDA next month, when the agency’s expert advisors will review EDAP’s Ablatherm-HIFU therapeutic ultrasound technology for treatment of prostate cancer.
EDAP’s losses widen, sales increase in 2012

EDAP (NSDQ:EDAP) posted a slight bump in sales but a dramatic increase in losses for the fiscal year of 2012
The French device maker posted year-end losses of $9.7 million, or 55¢ per diluted share, on sales of $33.7 million. That compares with 2011 losses of $1.3 million, or 10¢ per diluted share, on sales of $31.2 million.
FDA accepts Edap’s Ablatherm PMA application

Edap (NSDQ:EDAP) said the FDA accepted its pre-market approval application for its Ablatherm ultrasound prostate cancer treatment, backed by data from its Enlight investigational device exemption study.
The Vaulx-en-Velin, France-based medical device company also reported that its losses increased by a whopping 646% during the 2012, on an 8% sales increase.



