
California medtech company Direct Flow Medical notched some major milestones today, announcing a European regulatory win and completed enrollment in a U.S. feasibility trial for its transcatheter aortic valve implantation systems.
The Medical Device Business Journal — Medical Device News & Articles | MassDevice

California medtech company Direct Flow Medical notched some major milestones today, announcing a European regulatory win and completed enrollment in a U.S. feasibility trial for its transcatheter aortic valve implantation systems.

Direct Flow enrolled its 1st patient in a new U.S. clinical trial for the company’s transcatheter aortic valve implantation system, having earlier this year won FDA approval to launch the study. The study will treat a total of 30 patients from 6 clinical sites in the U.S., the 1st of which was enrolled at the Henry Ford Hospital in Detroit, Mich.
Corrected September 11, 2013, at 3:30 p.m.*

The FDA granted Direct Flow Medical an investigational device exemption for its transcatheter aortic valve implant, with the medical device company announcing enrollment in its Salus feasibility trial.

Being one of the smallest players on the field means you’ve got to differentiate yourself to stand out against the titans of the game, and that’s what Direct Flow Medical CEO Bernie Lyons aims to do.

California-based medical device maker Direct Flow Medical touted European regulatory approval for its transcatheter aortic valve implantation system, which the company says "virtually eliminates aortic regurgitation."
The device touts a metal-free frame and a low-profile delivery system, as well as a double-ring design that is both repositionable and retrievable after full deployment.

Medical device maker Direct Flow Medical unveiled 30-day results for its namesake transcatheter aortic valve implantation system, touting reduced valve leakage and procedure-related complications for the inflatable device.
In the study, which will enroll up to 100 patients, Direct Flow’s TAVI met primary mortality endpoints with zero incidents of major vascular complications and with 95% of patients experiencing mild or less paravalvular regurgitation.

It’s not your father’s transcatheter aortic valve implant anymore.
Next year and the year after, the competitive landscape for TAVI will explode, with as many as 12 competing devices on the market here and across the pond.
And that competition will be fierce. As TAVI celebrates its 10th birthday,at least 1 analyst is predicting a 20%+ compound annual growth rate for the market, which she says could potentially double by 2015.
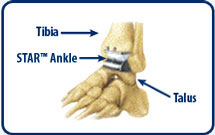 Star total ankle replacement
Star total ankle replacementOrthopedic technology company Small Bone Innovations Inc. landed a $43 million loan to further sales of its flagship ankle replacement device.
The New York-based company plans to use the credit facility as working capital to drive U.S. growth of its Star total ankle replacement, a non-cemented implant designed to replace arthritic ankle joints in patients with osteoarthritis, post-traumatic arthritis or rheumatoid arthritis.
The system is currently the only mobile-bearing total ankle replacement approved for the U.S. market.

Medtronic Inc. (NYSE:MDT) named GE Healthcare’s former chief, Omar Ishrak, to take over from chairman and CEO William Hawkins, effective June 13.
The news from the Fridley, Minn.-based medical device behemoth came just one day after rival Boston Scientific Corp. (NYSE:BSX) announced that CEO J. Raymond Elliott will be retiring at the end of the year.

