The U.S. Dept. of Homeland Security (DHS) issued a medical advisory warning for a potential software vulnerability in the Pyxis MedStation and Pyxis Anesthesia (PAS) ES system made by Becton Dickinson (NYSE:BDX). According to the DHS warning, Becton Dickinson reported the vulnerability to the Cybersecurity and Infrastructure Security Agency (CISA). Affected models of the drug-dispensing devices […]
Dept. of Homeland Security
DHS warns on Insulet Omnipod
The U.S. Dept. of Homeland Security (DHS) issued a medical advisory to warn of vulnerabilities with the Insulet (NSDQ:PODD) Omnipod insulin management system. According to the advisory, the affected insulin pumps are designed to communicate using a wireless RF with Insulet’s personal diabetes manager device, but the RF communication protocol does not properly implement authentication or […]
FDA, DHS ink cooperative medtech cybersecurity agreement
The FDA said yesterday that it is partnering with the U.S. Department of Homeland Security seeking to jointly improve cybersecurity in medical devices. The two agencies inked a memorandum of agreement looking to implement a new framework that will improve coordination and cooperation between the two bodies, according to the release. Through the agreement, both […]
DHS official: Ransomware an increasing threat to medical device cybersecurity
Medical devices will increasingly be the target of ransomware, according to a US Department of Homeland Security official. Threats to medical devices are expanding alongside the expansion of Internet of Things technology, according to DHS Industrial Control Systems Cyber Emergency Response Team director Marty Edwards. “It’s only a matter of time before we see some […]
Obama administration taps new anti-hacking agency
U.S. government probes medical devices for possible cyber flaws
Hacking: Password risk affects some 300 medical devices, says Homeland Security

The Dept. of Homeland Security warned last month that some 300 medical devices may be vulnerable to malicious hacking thanks to manufacturers’ hard-coded default passwords. Devices at risk include external defibrillators, infusion pumps, lab and analysis equipment, ventilators and more.
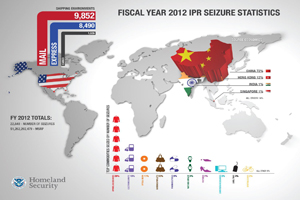
U.S. Customs seized $83M worth of counterfeit medical devices, drugs in 2012
MassDevice.com +3 | The top 3 med-tech stories for May 24, 2012.

Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
Device hacking: Federal agencies urge heightened review of device cybersecurity

A handful of government bodies are asking federal regulators to establish medical device software security protocols as part of the FDA’s regular review of medical devices.
The U.S. Dept. of Homeland Security joined the Information Security & Privacy Advisory Board of the National Institute of Standards & Technology in calling for heightened review of medical devices, citing security weaknesses that may leave them vulnerable to "malicious tampering."