SuperSonic Imagine said today its CEO Tom Egelund, only appointed as chief in April, will be replaced by supervisory board member Bernard Doorenbos who will operate as interim president and CEO. Doorenbos has held positions at Philips, Thomson CSF, Signaal USFA and acted as CEO of molds and plastic component company Axxicon Group NV, Neways Electronics […]
CVRx Inc.
FDA puts CVRx’s BaroStim heart failure treatment on the fast track
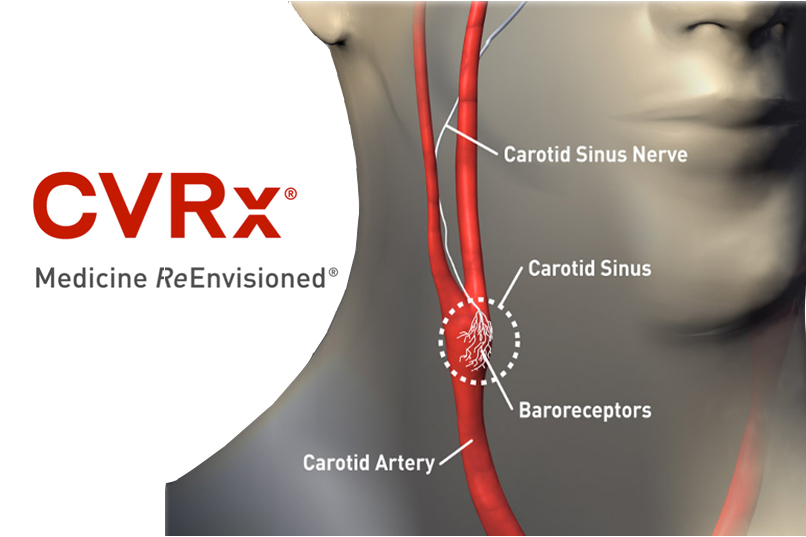
CVRx said today that it won a fast-track designation from the FDA that could shave 1 or 2 years from the timeline to approval for its Barostim Neo heart failure treatment. The Barostim Neo device is a neurostimulation implant designed to treat heart failure by influencing both the sympathetic and parasympathetic nervous systems, reducing sympathetic activity […]
HRS 2015: Boston Scientific, St. Jude Medical come out looking good
HRS 2015: CVRx’s Barostim headed for U.S. pivotal trial
The principal investigator in an early feasibility study of CVRx‘s Barostim device said today that the company won FDA approval for a pivotal trial later this year, after reporting strong results from an early feasibility trial of the device.
LifeScience Alley’s new chairman is CVRx CTO Kieval
LifeScience Alley, the Minneapolis-based trade association of medical device and life science companies, has chosen a new board chair and made other changes to its board of directors.
FDA awards priority review status to Cohera Medical’s surgical sealant Sylys | Regulatory news for the week of December 22, 2014
Hypertension: CVRx wins FDA nod for Barostim neo legacy
CVRx’s Barostim neo gets CE Mark for use with MRIs
CVRx said its Barostim neo system was granted expanded CE Mark approval in the European Union for use with MRI procedures.
CVRx wins heart failure CE Mark for Barostim Neo neurostim device
CVRx said today that it won CE Mark approval in the European Union for its Barostim Neo device to treat heart failure patients using vagus nerve stimulation.
Podcast: Medical device execs react to Medtronic-Covidien deal, M&A activity at DeviceTalks Minnesota

DaVita gets in on $12M round for CVRx

CVRx said it added $12 million from DaVita HealthCare, Ysios Capital and others to its Series F funding round, taking the total for the round to $42 million.
The Minneapolis-based company is planning to use the money to advance clinical trials and expand its international commercial footprint, according to a press release. CVRx developed the Barostim Neo, a pacemaker-like device designed to treat hypertension.