(Reuters) — Coloplast (CPH:COLO B) reported a 16% increase in 2nd-quarter core profit, slightly below expectations, pushing the medical device company’s share price down nearly 4%.
Coloplast
Pelvic mesh lawsuits: Judge urges plaintiffs, medtech companies to settle
Pelvic mesh lawsuits push Coloplast’s annual profits down
Philly court sets up new pelvic mesh mass tort
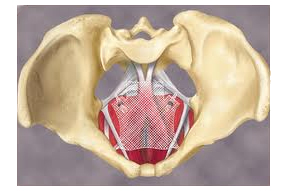
A Pennsylvania state judge ordered the creation of a mass tort this week to collect product liability lawsuits filed over pelvic mesh products against some of the largest names in medical devices.
Coloplast gains on Q4, FY2013 results
First bellwether date set for Boston Scientific in pelvic mesh lawsuits

The 1st bellwether trial in the multi-district litigation filed over Boston Scientific‘s (NYSE:BSX) pelvic mesh products is slated for early next year, a federal judge ordered this week.
Pelvic mesh makers look to settle lawsuits
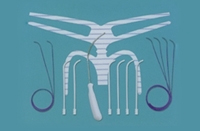
Five medical device companies that make pelvic mesh implants are hoping to negotiate settlements for the more than 30,000 pending lawsuits they’re facing in the U.S., anonymous sources told Bloomberg.
French pelvic mesh maker dodges Coloplast personal injury lawsuit

The French manufacturer of the mesh used in Coloplast’s embattled Aris transobturator tape system was dismissed from one of the thousands of personal injury lawsuits filed over pelvic mesh systems.
Coloplast can’t shake personal injury suit over urethral sling
Intuitive battles consolidation of da Vinci injury lawsuits | Legal News
Agilent’s $2.2B all-cash bid for Dako is largest in company history | MassDevice.com On Call

MASSDEVICE ON CALL — Agilent Technologies (NYSE:A) doled out more than $2 billion in cash to acquire Dako, a Denmark-based cancer diagnostic company with more than 1,000 employees with a foothold in more than 100 countries.
The deal, intended to grow Agilent’s life sciences business, is the largest in the company’s history, according to a press release.





