Biotronik
HRS 2015: Biotronik’s Iforia ICD safe for MRI
Biotronik‘s Iforia ProMRI implantable cardioverter defibrillator is safe for use in 1.5T MRI scanners, researchers announced today at the Heart Rhythm Society’s annual meeting in Boston.
Biotronik looks to get into the TAVI game
Biotronik today said its entry into a hot replacement heart valve market passed the safety endpoint of a 1st-in-human trial.
The German medical device giant said the Biovalve trial of its transcatheter aortic valve implant established the device’s early safety at 30 days.
Biotronik wins FDA nod for high-energy ICD
Biotronik said last week that the FDA approved 2 of its implantable cardioverter defibrillators designed to deliver an ultra-high-energy initial shock.
On-X Life Technologies wins expanded labeling indication from FDA | Regulatory news for the week of April 6, 2015
Olympus agrees to pay $92m to settle accounting scandal | Medtech legal news for the week of April 6, 2015
Biotronik launches Pantera Pro balloon catheter in Europe
Biotronik today said it’s launching the Pantera Pro balloon catheter, which won CE Mark approval in the European Union in January.
Insurer wants out of $28m Biotronik legal loss
An insurance company that issued policies to Biotronik wants a federal judge in Oregon to declare that they don’t have to cover Biotronik’s losses in a pair of lawsuits filed over allegedly unneeded pacemaker implantations.
BSX wins CE Mark, FDA nod for new S-ICD | Regulatory news for the week of March 23, 2015
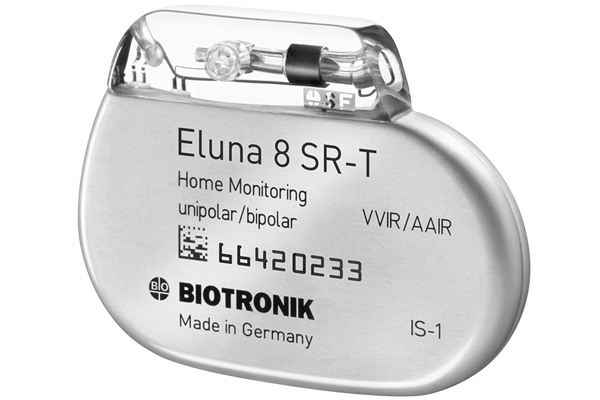
FDA approves Biotronik’s Eluna MRI-safe pacemaker
Biotronik said today that the FDA approved its ProMRI Eluna MRI-safe pacemaker, making it the only company with single- and dual-chamber MRI-safe pacemakers on the U.S. market.