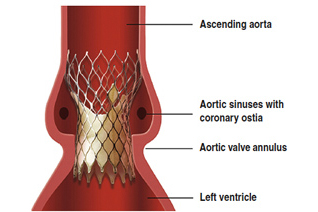
(Reuters) — Transcatheter aortic valve implantation is safe for low-risk patients, according to a study presented at the American College of Cardiology meeting in San Diego.
American College of Cardiology
ACC 2015: Medtronic readies ‘drug-filled stent’ trial
ACC 2015: St. Jude Medical’s CardioMEMS HF lowers mortality, hospitalizations
TAVI: Strong results for heart valves from Edwards Lifesciences, Medtronic
Boston Scientific’s Watchman misses 1 of 2 efficacy goals in PREVAIL trial
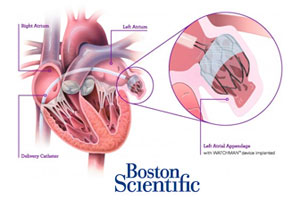
Boston Scientific (NYSE:BSX) this week unveiled full results from the PREVAIL clinical trial of the Watchman anti-stroke implant, largely agreeing with earlier reports that the device missed a co-primary endpoint for efficacy.
Bakris: Symplicity-3 a prologue for renal denervation | MassDevice.com On Call
ACC14: The quickening pace of change

I just returned from the American College of Cardiology and wanted to write down a few of my 50,000-foot impressions from the meeting for, as the song goes, the times, they are a-changin’.
Analyst speculates Smith & Nephew split | Medtech Wall Street news for the week of Mar. 31, 2014
ACC 2014 Roundup: Study shows long-term benefits for some CRT-D patients

Some patients with mild heart failure show significant, long-term benefits from cardiac-resynchronization therapy, according to the 7-year results from Boston Scientific‘s (NYSE:BSX) MADIT-CRT clinical trial presented at the American College of Cardiology’s annual meeting over the weekend.
ACC 2014 UPDATE: Reaction to Medtronic’s Symplicity renal denervation data
ACC 2014: Sapien XT bests CoreValve in small head-to-head study

UPDATED March 31, 2014, with comment from Medtronic