
BSX loses $309M judgment in patent war with the family of Dr. Michel Mirowski | Medtech legal news for the week of September 29, 2014

The Medical Device Business Journal — Medical Device News & Articles | MassDevice


Irvine, CA – AcuFocus, Inc. (www.acufocus.com), the global leader in small-aperture technology, today announced that its extended depth of focus intraocular lens (IOL), the IC-8™ small aperture IOL, has achieved CE Mark (Conformité Européenne) approval in Europe. The lens is designed to provide exceptional visual quality across a broad range of vision for presbyopic patients with cataracts.
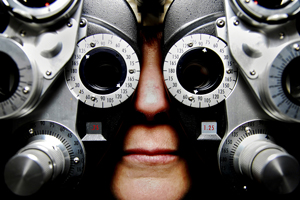
California-based device maker AcuFocus is taking its next-generation intraocular lens to Europe after winning regulatory approval to treat cataract patients with near vision loss, or presbyopia.
AcuFocus said today that it raised another $21 million in an oversubscribed round as it awaits a decision from the FDA on its Kamra device, a corneal inlay designed to treat near vision loss, or presbyopia.

A panel of FDA experts today was divided on whether the federal watchdog agency should approve the AcuFocus Kamra Inlay, a 1st-of-a-kind permanent corneal implant for the improvement of near and intermediate vision in patients with presbyopia, a form of age-related far-sightedness.

The FDA said it plans to hold an advisory panel hearing to evaluate the Kamra Inlay device made by AcuFocus to treat presbyopia, or near vision loss.
The FDA’s Ophthalmic Devices Panel will convene June 6 to consider AcuFocus’ bid for pre-market approval for the Kamra device, a ring-shaped inlay that’s implanted under the surface of the cornea, the federal watchdog agency said today.

Insulet (NSDQ:PODD) said it named Dr. Howard Zisser, the former clinical director of the Sansum Diabetes Research Institute, as its 1st medical director.
Considered a leader in diabetes research, Zisser will be involved with a variety of clinical research projects and serve as a medical liaison between Insulet and government leaders, the FDA, and healthcare organizations, Bedford, Mass.-based Insulet said.

James Mazzo has put 35 years into the optics and ophthalmology business, so it was no surprise that he got involved with AcuFocus and its device for treating presbyopia. Mazzo, who stepped down from leading Abbott Medical Optics earlier this year, added another title to his resume that’s more of a departure: He’s the 1st "operating partner" at Versant Ventures.

Johnson & Johnson (NYSE:JNJ) Ethicon Endo-Surgery won FDA clearance for the 1st surgical instruments that can be assembled and disassembled inside the body during surgery.

