The company commenced enrollment at the University of Pittsburgh in Pittsburgh, Pennsylvania. It plans for its early feasibility study to primarily assess safety while exploring quantified efficacy measures of the Synchron Stentrode.
Synchron’s study, funded by the National Institutes of Health (NIH), involves patients with severe paralysis. It aims to enable the patient to control digital devices, hands-free.
READ: 7 brain-computer interface companies you need to know
The study operates under an FDA investigational device exemption (IDE). Synchron said in a news release that it marks the first such IDE for assessing a permanently implanted BCI. The new site joins Mount Sinai (New York) as the second approved COMMAND trial location.
“Today marks an important milestone in our mission to advance technology access for the millions of people who have lost the ability to use digital devices,” said Dr. Tom Oxley, CEO & founder, Synchron. “We are excited to expand the COMMAND trial with enrollment at the University as we look to advance a scalable BCI solution to market.”
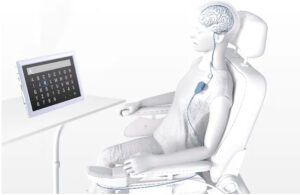
About Stentrode
Stentrode is implanted within the motor cortex of the brain through the jugular vein in a minimally invasive endovascular procedure. The endovascular BCI approach differs from competitors as it does not require invasive open-brain surgery.
Once implanted, it detects and wirelessly transmits motor intent. This restores a capability for severely paralyzed patients to control personal devices through hands-free point-and-click.
The company said its trial assesses the impact of tasks like texting, emailing, online shopping, and telehealth services.
“This exciting technology may open up the possibility for patients with paralysis to use digital devices to communicate with loved ones again, and restore independence back into their lives,” said Dr. David Lacomis, principal investigator and chief of the Neuromuscular Division at the University of Pittsburgh. “Something as simple as surfing the web or doing online grocery shopping can seem mundane to some, but it can change the world for someone who previously had those fundamental abilities taken away.”
BCI space continues to heat up
In August, Reuters reported that Elon Musk approached Synchron over a potential investment.
Musk’s Neuralink BCI company fell behind Synchron this past summer. This came after Synchron completed its first-in-human brain-computer interface (BCI) implant in the U.S.
Neuralink is developing an implant placed in the brain through a robot-assisted procedure. While Musk and company officials said they planned to file for FDA approval for human trials in 2020, it has yet to receive any such approval. Earlier this year, Neuralink and the University of California, Davis, were accused of “egregious violations of the Animal Welfare Act” by the Physicians Committee for Responsible Medicine (PCRM), citing documents obtained through a public records lawsuit. The allegations claimed that Neuralink caused extreme suffering in monkeys.
Musk scheduled a “show and tell” for Neuralink on Oct. 31 of this year. However, he pushed the event back a month for unknown reasons, per Reuters.
Synchron and Neuralink are not alone in the BCI space. In fact, Blackrock Neurotech’s Utah Array has been used in patients since 2004 through research studies, with zero FDA-reported serious adverse events since then. (Check out our roundup of BCI companies you need to know. )

