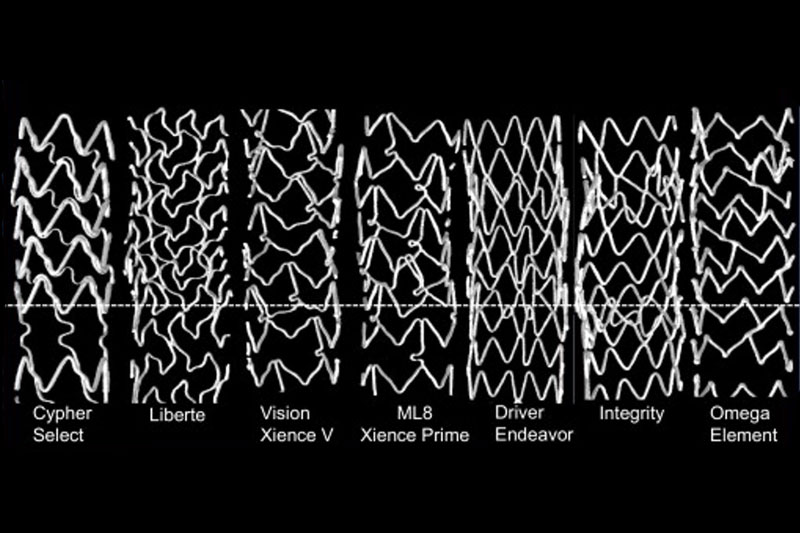
Some stents made by Boston Scientific (NYSE:BSX) and Medtronic (NYSE:MDT) fared worse in compression tests than competing designs, a new study found.
In a bench-test study of seven different stents, Medtronic’s Driver and Boston Scientific’s Omega stent warped under significantly less force than other models.
The results add to mounting concerns that the thinner design of next-generation coronary stents may be prone to crumpling after deployment.
Johnson & Johnson (NYSE:JNJ) subsidiary Cordis Corp.’s Cypher stent proved the strongest of the seven stents tested. Cordis announced over the summer that plans to pull out of the stent business entirely and stop making the Cypher line by the end of the year.
Researchers also tested Boston Scientific’s Liberté, Medtronic’s Integrity and Abbott’s (NYSE:ABT) Vision and MultiLink devices.
Under 0.5 N of force, Omega shrank 5mm, Driver shrank 4mm, Liberté and Integrity each shrank 2mm, Vision and MultiLink each lost 1mm and the Cypher didn’t warp at all.
To achieve 5mm of shrinkage, Driver required 0.71 N of force, Integrity required 1.08 N, Liberté and MultiLink required 1.10 N, Vision required 1.12 N and Cypher required 1.33 N.
Boston Scientific’s Promus Element stent, which landed FDA clearance just last week, was the third-weakest tested.
At least two studies and one case study presented last month at the Transcatheter Cardiovascular Therapies conference in San Francisco reported incidents in which either a Boston Scientific Ion or Promus stent deformed after it was deployed inside a coronary artery. The FDA has taken an interest in the concerns, launching an investigation into newer stent models.
"FDA is actively working with manufacturers, including Boston Scientific, to better understand longitudinal stent deformation with respect to its causes, predisposing underlying anatomic conditions, operator techniques that can reduce the likelihood of its occurrence, and treatment strategies should it occur," according to the agency.
Although there’s been only one reported incident of deformation involving a Promus stent from among more than 4,600 implantations in clinical trials, according to the FDA, "an unspecified number of significant adverse events had been reported to the agency."
"At this time, additional data collection and analyses are ongoing, but the information available to date indicates that the Ion paclitaxel-eluting platinum chromium stent remains safe and effective when used for its approved indications," the FDA said, according to Reuters.
An Irish study published earlier this month found that thinner stents may have lower longitudinal strength, potentially leading to stent thrombosis and a "catastrophic late complication" and leaving the target lesion unprotected, according to scientists at the Belfast Health & Social Care trust.
"When struts of a deployed stent are pushed together or pulled apart, longitudinally deforming a stent, there are regions of artery that are no longer fully scaffolded and where drug delivery may be reduced, with a potential for restenosis," according to the new study.
If determined to be a flaw, the news could affect the $4 billion stent market. That would leave Boston Scientific, which gets 20 percent of its revenues from drug-eluting stents, especially vulnerable, Wells Fargo Securities analyst Larry Biegelsen wrote in a note to investors.
Stent shrinkage is relatively rare compared to other problems associated with clearing blocked vessels, Boston Scientific interim CEO Hank Kucheman told investors during the company’s third-quarter conference call.
"Stents have a lot of different properties, which vary from one to another," New York Presbyterian Hospital and Colombia University Medical Center director of cardiovascular research Dr. Gregg Stone said during a conference call discussing the Belfast report. "Some stents will be more prone to this complication than others. The question is to understand how frequent it is, when does it occur, how to prevent it and how to manage it."