
Say hello to MassDevice +3, a bite-sized view of the top three med-tech stories of the day. This latest feature of MassDevice.com’s coverage highlights our three biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else today, make sure you’re still in the know with MassDevice +3.
3. FDA releases two new graft guidances

The FDA released two draft guidances for products under investigational device exemptions that wish to embark on human testing and updates its standard operating procedure for requesting new data on pre-market submissions.
![]()
Sign up to get our free newsletters delivered straight to your inbox
![]()
2. Docs with MDT ties refuse to disclose potential conflicts
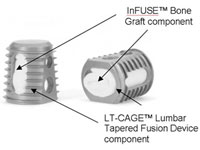
Doctors accused of withholding information regarding the potential risks associated with Medtronic’s (NYSE:MDT) Infuse bone growth protein refuse to disclose the amount of money they received from the med-tech titan in the years the studies were published.
![]()
1. Stryker plans to lay off 5% of workforce ahead of med-tech tax

Stryker Corp. (NYSE:SYK) officials said the company plans to cut 5 percent of its workforce over concerns about the impending 2.3 percent medical device tax prescribed by President Barack Obama’s health care overhaul.

