 The battery depletion issue confronting St. Jude Medical (NYSE:STJ) this week is only the latest such issue to bedevil a medical device maker, as its arch-rivals in the cardiac rhythm management space have issued their own recalls in recent years.
The battery depletion issue confronting St. Jude Medical (NYSE:STJ) this week is only the latest such issue to bedevil a medical device maker, as its arch-rivals in the cardiac rhythm management space have issued their own recalls in recent years.
St. Jude yesterday warned of the possibility that a battery issue could disable its high-voltage cardiac rhythm management devices, saying the problem is associated with 2 deaths among the nearly 400,000 patients implanted with affected devices. The news sent STJ shares down -3.5% to $78.41 apiece yesterday and the stock was down another -0.1% to $78.30 today in mid-morning trading – partially erasing its recovery from a short-seller’s allegation of cybersecurity vulnerabilities with its Merlin@home remote monitoring system.
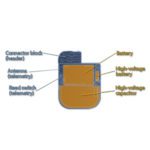
The problem stems from a risk that the lithium-based batteries used in its implantable cardioverter defibrillators and cardiac resynchronization therapy devices could form “lithium clusters” during high-voltage charging. The clusters could then cause a short circuit and deplete the battery within a day to a few weeks, rendering the device incapable of delivering therapy. Dr. Mark Carlson, chief medical officer at St. Jude, said most patients won’t require prophylactic replacement “because the risk associated with replacing the devices outweighs the low risk of a patient problem occurring.”
As far back as 2012, St. Jude’s CRM competitors dealt with similar issues. In March of that year, Medtronic (NYSE:MDT) issued an alert on its EnTrust and and Escudo ICDs, warning that the devices may be susceptible to an internal short circuit that could drain the battery. The following year, Boston Scientific (NYSE:BSX) said its by-then-discontinued Cognis and Teligen implantable cardioverter defibrillators might be subject a component issue that could deplete the battery entirely after 2 years. And as recently as last month, Medtronic said a small number of its Viva CRT-Ds and Evera ICDs might have a low resistance path that could result in rapid battery depletion.
Neither company showed any adverse financial effects from the battery issues, according to Barclays analyst Matt Taylor. Wedbush Securities analyst Tao Levy said the recall would likely cost St. Jude no more than “a few million dollars” because relatively few patients will need early replacements and devices now being sold have improved and reliable batteries.
And although investors yesterday also punished both Medtronic and Boston Scientific (MDT shares closed off -3.2% at $83.20 each, while BSX shares closed down -1.9% at $23.16 apiece), both stocks recovered somewhat today, rising 0.5% to $83.62 and 0.4% to $23.25, respectively, in mid-morning activity.
For its part, Medtronic sought to distance itself from St. Jude’s woes.
“[Medtronic] uses a different battery, with a different battery design,” a spokeswoman told Benzinga. “Also, we manufacture our own batteries.”
Abbott (NYSE:ABT), which is waiting on European anti-trust regulators to rule on its pending, $25 billion acquisition of St. Jude, said the latest hurdle hasn’t dampened its ardor.
The Abbott Park, Ill.-based company still expects its acquisition of St. Jude to close by the end of this year, spokesman Scott Stoffel told the Chicago Tribune via email.
Material from Reuters was used in this report.

