Medtronic (NYSE:MDT) had to continue its Spyral HTN-ON MED trial into this year after failing to obtain the positive results needed to end enrollment early for the potential hypertension treatment.
During Medtronic’s Q3 earnings call in February, CEO Geoff Martha said the company expects to complete the six-month follow-up to ON MED in the second half of this calendar year. It’s the final piece of information Medtronic needs to submit for FDA approval of its Symplicity Spyral device.
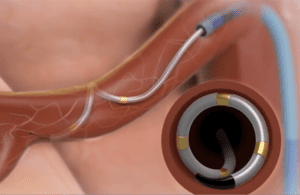
Medtronic’s Symplicity Spyral device delivers energy to the nerves in the wall of the artery leading to the kidney during a minimally invasive outpatient procedure. The idea is to calm overactivity in the nerves that can cause high blood pressure.
Get the full story on our sister site Medical Design & Outsourcing.

