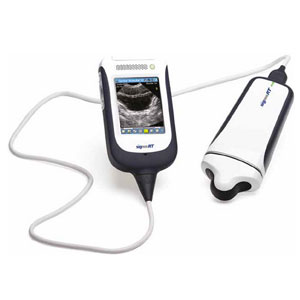
Australian medical device company Signostics won 510(k) approval for its handheld ultrasound device.
The device will be sold as the Sonimage P3 through the its partnership with Konica Minolta (TYO:4902). This device already is on the market in Europe and Australia, where it is sold as Signos RT.
Sonimage P3 is an update of a previous version that has been sold in the U.S. since 2009. The new version has a longer battery life and pulsed wave Doppler imaging. Sonimage P3 is a handheld and self-contained ultrasound, with the monitor display attached with a cord.
The device can save up to 1,000 images to an SD card and also comes with USB connectors. It is intended for quick uses, such as basic pregnancy assessment, bladder volumes and obvious pathology assessments.
“FDA clearance for the USA market further enhances the potential of this innovative product and will enable medical practitioners and patients throughout America to benefit from the real-time point-of-care imaging capabilities that the device provides,” said Signostics CEO Warren Ortmann in prepared remarks.

