The ShoulderFlex personal massager by King International that was recalled in September after the device killed one patient, nearly strangled another and injured two more is still on the market, according to the FDA.
During a recent compliance audit, the federal watchdog agency found that the company had gone out of business without following through with recall procedures.
The 800 number established for the recall is out of service and companies that sell the device and customers that purchased it might not be aware of the recall.
Sign up to get our free newsletters delivered straight to your inbox
"The ShoulderFlex Massager poses serious risks. Consumers should stop using this device, health care providers should not recommend it to their patients and businesses should stop distributing and selling the device," Steve Silverman, director of the Office of Compliance in the FDA’s Center for Devices & Radiological Health said in prepared remarks.
The federal watchdog agency upgraded the now-defunct personal massager maker’s voluntary September recall of the ShoulderFlex system to a Class I designation in November.
The federal watchdog agency’s Class I designation, the most series category, is applied to devices where there is a reasonable probability that the products will cause series health consequences or death.
Beaverton, Oreg.-based King distributed nearly 12,000 of the ShoulderFlex massagers through stores and online retailers throughout the U.S. since October 2003. Under terms of the recall, King International was supposed to notify all affected parties.
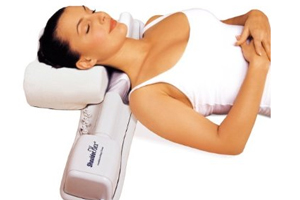
ShoulderFlex is a therapeutic massager that provides deep tissue massage to the neck, shoulder and back while a user lies on it. It contains a rotating bar with removable "fingers" that can be adjusted to modify the massage.
Massage experts recommend to buy massage chair to the office or home if you can afford it. Massage reduce your stress and muscle tightness. We did some research on massage chairs and found list of best massage chairs for 2018 with buyers guide, you definitely want to read the reviews before you buy one because the price range is wide and they have different functions.
The incidents leading to the warning and subsequent recall occurred when a necklace and piece of clothing got caught in the device’s rotating massager. The FDA noted that it had two other reports of hair and clothing being caught in the device.

