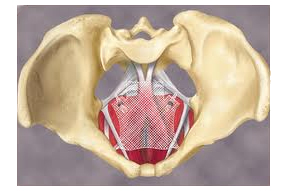
Allegations made by Johnson & Johnson (NYSE:JNJ) about a campaign of telephone solicitation aimed at generating more lawsuits over its pelvic mesh products aren’t backed by any proof, the plaintiffs argued last week, saying the company’s move to force the plaintiffs to prove they have valid claims is just a delaying tactic.
The company wants Judge Joseph Goodwin, who’s overseeing the multi-district litigation that covers 10s of thousands of product liability lawsuits, to force plaintiffs to prove that they have a legitimate claim, according to court documents. And it wants Goodwin to force the plaintiffs’ lawyers to testify under oath and in front of the judge "to determine who may be profiting from the unethical and illegal direct solicitation of women and whether further investigation of others is warranted," according to the documents.
The plaintiffs countered that there’s no proof that any of the lawsuits against Johnson & Johnson are fraudulent.
"Defendants have failed to establish that a single complaint out of 23,000-plus Ethicon cases is fraudulent. Not one. Indeed, independent random sampling – a survey governed by a protocol defendants developed – failed to reveal fraud in any filings," the plaintiffs claimed in a court filing. "Notwithstanding such clear evidence, defendants inferentially leap from citing the unscrupulous acts of a call center which, by defendants’ own admission, is likely unaffiliated with any of the plaintiff lawyers before this court, to the shocking claim that fraudulent filings are pervasive."
"Stripped of its anecdotal histrionics, defendants’ motion lacks any substance at all," according to the filing. "The absence of any established link between the call center’s actions and any plaintiff in this litigation means there is no basis for imposing any new discovery obligations on individual plaintiffs (much less on their lawyers)."
Johnson & Johnson’s Ethicon and Mentor subsidiaries aren’t the only medical device companies facing personal injury lawsuits over thier pelvic mesh offerings. C.R. Bard (NYSE:BCR), Boston Scientific (NYSE:BSX) and others are each facing thousands of the suits; those that have made it to trial have largely gone the plaintiffs’ way.
Last weekJ&J agreed to settle another batch of cases filed against Mentor. In November 2014, a jury in West Virginia awarded 4 women $18.5 million for injuries they said were caused by Boston Scientific‘s (NYSE:BSX) Obtryx device for stress urinary incontinence, including $4 million for "gross negligence." That verdict came a week after a Miami jury awarded $26.7 million to 4 women implanted with the company’s Pinnacle device for pelvic organ prolapse.
Bard, whichearlier this month lost a bid to delay some of its trials, last October inked a settlement deal for some 500 of the cases that’s reportedly worth about $21 million.