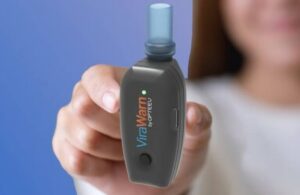
Baltimore-based Opteev designed its analyzer to detect COVID-19, influenza and RSV with results in less than 60 seconds.
It aims for the multiple-use, rechargeable analyzer to become an affordable diagnostic option. The company plans to unveil the device at CES Unveiled in Las Vegas today. It also intends to have the device on display at CES between Jan. 5-8.
Opteev said in a news release that it submitted ViraWarn to the FDA this past summer. It remains unavailable for marketing in the U.S.
The device uses a silk-based biosensor that attracts the electrical discharge of respiratory viruses. This, combined with an artificial intelligence processor, filters out potential inaccuracies. Users turn ViraWarn on and blow twice into the mouthpiece. An LED notification light then indicates a positive or negative result in under 60 seconds.
Opteev said that ViraWarn is reusable and comes with multiple biosensor replacement cartridges. Those only require replacement after a positive result or after a period of 2-3 weeks of daily usage.
“We’re looking forward to debuting ViraWarn to the public at CES this week,” said Conrad Bessemer, president and co-founder of Opteev. “ViraWarn is designed to allow users an ultra-fast and convenient way to know if they’re spreading a dangerous respiratory virus so they can stop the spread and save lives in the process. As COVID, RSV, and Influenza are continuing to surge, we’re eager to bring ViraWarn to market so consumers can easily blow into a personal device and immediately find out if they’re positive or negative.”

