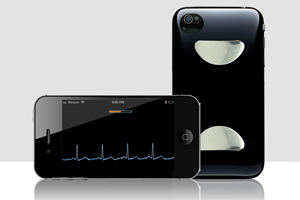
Utah-based Cardiac Designs is still flying under the radar after having won FDA 510(k) clearance for its iPhone-based ECG monitor.
The FDA nod may represent a win for consumers who are increasingly interested in recording their own health data, but the regulatory win comes with a strange restriction – patients must obtain a physician prescription in order to view the raw wave form data captured from the heart.
The device is not yet on the market and the company hasn’t announced its regulatory win, and the company website has little information beyond noting that the ECG Check iPhone cardiac monitor is "coming soon."
The raw data restriction is one that rival iPhone ECG company AliveCor aims to tackle as well, although how it plans to overcome it is "an AliveCor secret," a company spokesman told MobiHealthNews
AliveCor’s AliveECG heart monitor has garnered more public attention, especially since a cardiologist last week used the device to diagnose an abnormal heart rhythm mid-flight on a commercial airplane. It was the 2nd time the device had made headlines for a mid-air diagnostic, the 1st time occurring in 2011 when the device was used to diagnose a heart attack, spurring an emergency landing for the airplane.
AliveCor’s device isn’t FDA cleared for over-the-counter sales, but a physician can prescribe the device for patient use. The company hopes to gain OTC indication this year, according to MobiHealthNews

