Boston Scientific (NYSE:BSX) said today that it’s closed enrollment for the Prevail study of its Watchman heart implant with a final burst of 407 patients across 42 sites.
The randomized trial will support the medical device company’s bid for FDA clearance, but BSX will need to gather 6 months of data before submitting its application.
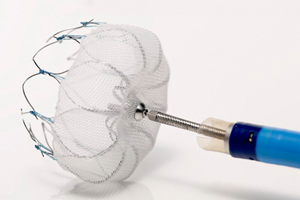
Incoming CEO Michael Mahoney – currently listed as president – forecast 2013 FDA approval and U.S. launch for the Watchman left atrial appendage closure device, which is designed to trap blood clots and may be an important tool in preventing stroke in high-risk atrial fibrillation patients.
"Watchman is the most clinically studied device of its kind," Mount Sinai Medical Center cardiac arrhythmia director Dr. Vivek Reddy said in prepared remarks. "Watchman has the potential to provide atrial fibrillation patients with a safe and effective first-in-class device-based solution to reduce risk of stroke."
The study, which began in November 2010, has enrolled more than 2,000 patients and gathered more than 4,000 patient-years of follow-up, according to a press release.
The trial’s 2 arms compared the Watchman implant with the anticoagulant drug warfarin in high-risk patients with atrial fibrillation
In the multi-center randomized Protect AF arm, Boston Scientific reported a 38% risk reduction for stroke, cardiovascular death and systemic embolism compared to long-term Warfarin therapy in 707 enrolled patients.
The prospective multi-center ASAP trial, the results of which were unveiled earlier this year at the Heart Rhythm Society meeting in Boston, found a 77% reduction in ischemic stroke risk associated with the device in patients who can’t tolerate the drug.
Asked if the Watchman device is a game-changer or just another tool in the electrophysiologist’s armamentarium, lead author Dr. Reddy of the Mount Sinai School of Medicine in New York told MassDevice.com that more studies are needed comparing the device with newer anticoagulants such as dabigatran.
"If the world only had Warfarin and the Watchman, I would say game-changing. As it is right now, I would say it’s an alternative. Right now we have drugs that are better than Warfarin and devices – I would say, that are better than Warfarin," Reddy told us at the HRS conference in May. "So the question then is which of these do we use, or which in combination? You can’t say it’s game-changing unless you prove that 1 is better than the other. Basically, we don’t know which is better. That kind of a study, comparing the new drugs with appendage closure, those are future studies that need to be done."
The Watchman device, which is already available in 30 countries, is introduced via catheter and threaded into the heart to close off the left atrial appendage and capture any blood clots that may form there. The device aims to reduce the risk of stroke and may potentially eliminate the need for long-term use of blood-thinning medications, according to a press release.

