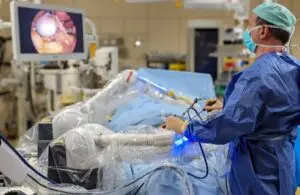
It’s more good news for the French company, which last week won FDA clearance for Maestro. The clearance offers support to surgeons in soft tissue surgical procedures.
Moon Surgical designed Maestro for soft tissue procedures not currently supported by telerobotic systems. The small, adaptable system can integrate into existing clinical workflows and provide the surgeon with more control.
The Maestro system is designed to increase operating room efficiency by enhancing surgical instruments and techniques and minimizing the impact on operating room turnover time.
Dr. Guy-Bernard Cadière performed all 30 procedures over 10 days at CHU Saint-Pierre, a public university hospital in Brussels, Belgium.
“I am thrilled to be part of this monumental accomplishment by Moon Surgical,” said Cadière. “This technology is brilliant; it gave me more control throughout the procedure, keeping me confident and less fatigued by the end. Maestro is a great benefit for any surgeon, giving them the freedom to conduct each surgery in the most efficient manner.”
Moon Surgical featured in MassDevice’s list of 16 surgical robotics companies you need to know
More on the procedures
The procedures covered seven different clinical indications, demonstrating Maestro’s adaptability in diverse procedures. Each procedure demonstrated that surgeons did not need a surgical assistant to hold and manipulate instrumentation.
Moon Surgical said 14 of the procedures were laparoscopic cholecystectomies (removal of the gallbladder to treat symptomatic gallstone disease). Others included bariatric interventions, hernia repairs, colorectal surgeries and gastric reflux surgery.
“This is another testament to how much the Maestro System can improve a surgeon’s experience,” said Anne Osdoit, CEO of Moon Surgical, and a partner in Sofinnova Partners’ medtech accelerator, MD Start. “The results of our first-in-human trial are a great validation of the value of the Maestro System in a real-life surgical setting. It is simply fantastic to see our system providing treatments to patients. There is no greater reward than this.”

