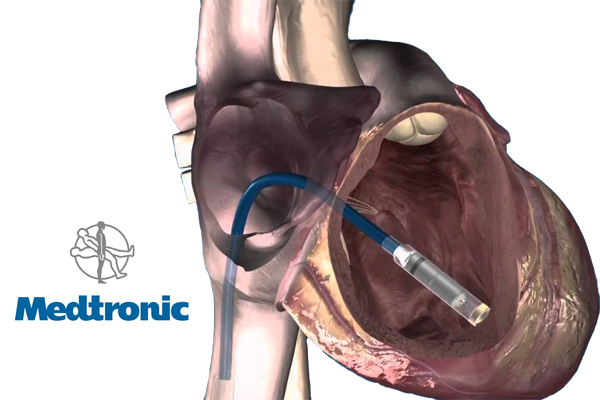
Medtronic‘s (NYSE:MDT) entry into the leadless pacemaker market won CE Mark approval in the European Union, the company said today, touting it as the world’s smallest pacemaker.
The Micra transcatheter pacing system, which at ⅒ the size of a conventional pacemaker is roughly the size of a large vitamin, is designed to be implanted via catheter in the right ventricle to deliver single-chamber pacing, Medtronic said.
"Unlike traditional pacemakers, the Micra TPS does not require leads or a surgical ‘pocket’ under the skin, so potential sources of complications are eliminated – as are any visible signs of the device," Dr. Philippe Ritter of France’s Hôpital Cardiologique de Haut Lévêque and Centre Hospitalier Universitaire Bordeaux said in prepared remarks. "While the Micra TPS is dramatically smaller, it is a fully self-contained pacemaker that still delivers the most advanced pacing technology available to patients."
The Micra device has an estimated 10-year battery life and is approved as safe for full-body MRI scans, Fridley, Minn.-based Medtronic said.
The CE Mark was granted based on 3-month results from the 1st 60 patients enrolled in Medtronic’s Micra TPS trial, the company said. The study is slated to enroll up to 720 more patients; initial results are due to be presented at the annual Heart Rhythm Society meeting in Boston this May, according to Medtronic.
"Our cross-functional teams have been working for years to redefine engineering limits and production capabilities by radically reducing the size of medical devices by more than 90% while continuing to innovate upon the existing technology," vice president Brian Urke said in a press release. "We believe our investment in this research is transforming cardiac care and will provide more and better therapy options to patients."
Cardiac rhythm management rivals St. Jude Medical (NYSE:STJ) and Boston Scientific (NYSE:BSX) also have leadless pacemakers in the works, although Boston’s S-ICD uses subcutaneous leads and a surgical pocket for the device. St. Jude’s Nanostim device, like the Micra, is designed to be implanted via catheter. The Nanostim device has been approved in Europe since 2013.