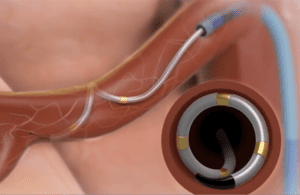
The highly anticipated results will offer new insight into the Fridley, Minnesota-based medical device manufacturer’s quest to build a billion-dollar business treating hypertension. CEO Geoff Martha recently reported “good progress” in those efforts, saying the trial “represents the final piece of a large body of evidence that we intend to submit to the FDA for approval.”

