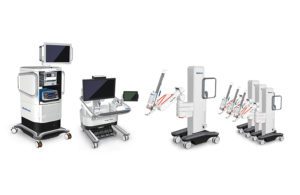
The Fridley, Minnesota-based company won CE mark clearance for general surgery indication, Health Canada license for general laparoscopic surgery indication and Ministry of Health, Labor and Welfare approval for urologic surgical and gynecological laparoscopic indications in Japan.
The new indications and approvals enable access to robotic-assisted surgery in Europe, North America and Asia to extend the benefits of minimally invasive surgery.
Robotic-assisted procedures have fewer complications, shorter hospital stays and faster return to normal activities. General surgery, gynecology and urology account for more than 80% of all robotic-assisted surgical procedures performed globally and general surgery is the fastest-growing segment within robotic surgery, according to Medtronic.
“Receiving these important approvals paves the way for our expansion into key robotic markets,” said Mike Marinaro, senior VP and president of surgical robotics at Medtronic. “The Hugo RAS system was designed to address the barriers to robotic surgery adoption, and these approvals are significant steps in bringing the benefits of minimally invasive surgery to more patients in more places around the world.”
Hugo uses the company’s Touch Surgery Enterprise, a cloud-based surgical video capture and management solution, to offer a smart, digitally enabled surgical experience. It has wristed instruments, 3D visualization and dedicated support teams that specialize in robotics program optimization, service and training.
“These regulatory approvals are a major step in expanding the benefits of minimally invasive surgery to patients around the world. We truly believe that AI-assisted robotic surgery will transform the operating room,” Chief Medical Officer of Surgical Robotics at Medtronic Carla Peron said in a news release. “Our team is thrilled to begin partnering with more surgeons and customers to help build and grow their robotic surgery programs in this hugely important area of healthcare.”

