Go directly to MassDevice. Having trouble reading this email? View this newsletter in your browser here.
Not interested? Unsubscribe.
The MassDevice Weekly Roundup brings you the latest medical device news and information.
The MassDevice Checkup
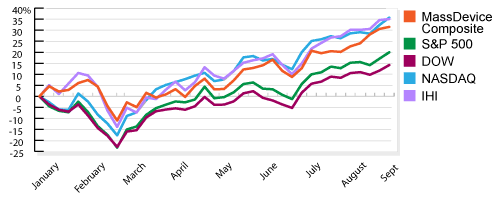
Our weekly checkup takes the temperature of the public medical device companies in the three largest markets in the U.S.: Massachusetts, Minnesota and California. In addition to the indices for each state, which track the overall movement of the sector, we compare Monday morning’s opening share prices with their value when the markets close Friday afternoon and calculate the biggest gainers and losers. Read More ![]()
![]()
News
Medtronic: Endeavor stent is safer than BoSci’s Taxus
The Minneapolis devices giant lobs another grenade at its Natick, Mass.-based rival in the stent wars. Read More ![]()
InVivo Therapeutics sues Oregon Health & Science University over primate study
The Cambridge, Mass.-based tissue regeneration developer accuses the research facility of mistakes it says have jeopardized its future. Read More ![]()
Boston Scientific, PerkinElmer release imaging software packages
The medical device leviathans début ultrasound and cellular imaging software packages. Read More ![]()
PrimeraDx lands $20 million Series C round
The molecular diagnostics maker drums up another hefty chunk of change from new and existing investors to advance its commercialization efforts. Read More ![]()
CardioFocus atrial ablation device gets first use in U.S. clinical trial
Physicians in New York used the Marlborough, Mass.-based company’s visually-guided laser balloon catheter in an atrial ablation procedure for the first time in a domestic clinical trial. Read More ![]()
Inverness gets healthy for $130 million in Free & Clear buyout
The Waltham, Mass.-based diagnostics maker buys Seattle’s Free & Clear, which provides programs aimed at improving health, for $100 million in cash and up to $30 million in revenue-pegged contingencies. Read More ![]()
FDA chief takes a hard look at glucose monitors
Food & Drug Administration commissioner Margaret Hamburg is pushing for tougher international standards for diabetes glucose monitors. Read More ![]()
Pressure BioSciences fills board seat
The pressure cycling device maker replaces the retiring Thomas Vogel with Alan Rosenson on its board of directors. Read More ![]()
BioSphere Medical settles lawsuit over blinded Missouri youth
Proposed settlement could avert a trial slated for next month seeking to assign responsibility to BioSphere’s microbead technology. Read More ![]()
Family scales back secondary offering of Bruker stock
A proposed deal by members of the founding family of the Billerica, Mass.-based maker of scientific equipment is cut by nearly two-thirds. Read More ![]()
Boston Scientific: 1,500 patients enrolled in Promus Element stent trial
The Natick, Mass.-based devices giant said the workhorse clinical trial of its Promus Element drug-eluting stent finished enrolling patients two months ahead of schedule. Read More ![]()
Wadsworth gets regulatory nod for DermaLOC wound closure
The Westborough, Mass.-based start-up will market an alternative to needles and staples for closing wounds and surgical incisions. Read More ![]()
Deerfield Management cashes in on NxStage
New York hedge fund reaps a small windfall from the renal products maker and ups its stake in NeuroMetrix. Read More ![]()
Kerry expresses concern over taxing the medical device industry
Massachusetts’ lone senator stops short of joining the Indiana and Minnesota delegations’ opposition to a proposed $4 billion annual tax on medical device makers. Read More ![]()
Shareholders slap CardioNet with lawsuits
Shareholders of the mobile cardiac monitoring firm are suing the company and its CEO and CFO, alleging that they misled investors over the company’s prospects for high Medicare reimbursement. Read More ![]()
 MIT’s Enterprise Forum launches medical device entrepreneurship program
MIT’s Enterprise Forum launches medical device entrepreneurship program
Nine-week program for would-be medical device entrepreneurs starts Sept. 22 in Newton. Read More ![]()
Helicos BioSciences raises $9.4 million in private placement
New funding round nearly doubles cash on hand for the Cambridge, Mass.-based DNA analysis equipment manufacturer. Read More ![]()
A mixed day for Medtronic
The Minneapolis device giant wins a patent lawsuit against W.L. Gore, but its recall of subsidiary Physio-Control’s Lifepak AED goes Class I. Read More ![]()
Cynosure hires former Candela exec
Paul Cardarelli, the cosmetic device maker’s new VP of business development, will lead its joint venture with Unilever to develop home-use devices. Read More ![]()
Haemonetics inks deal with Blood Centers of America
The community blood center cooperative will use the Braintree, Mass.-based firm’s blood management software. Read More ![]()
NMT Medical pushes up StarFlex trial
The Boston-based cardiac implant maker plans to begin analyzing trial data in April 2010, ahead of its fall target date, potentially setting the stage for a PMA application to the FDA in after the third quarter next year. Read More ![]()
Perceptive Informatics upgrades its image with Optasia Medical
Parexel International’s clinical trial data management arm will use Optasia’s KneeAnalyzer software for osteoarthritis studies. Read More ![]()
Optos taps Soden as interim CFO
The retinal imaging maker taps Christine Soden as interim CFO after the departure of Allan Watson. Read More ![]()
PerkinElmer buys GE Healthcare reagent lines
The Waltham, Mass.-based devices and diagnostics firm acquired the conglomerate’s catalog radiochemical, SPA reagent and Cytostar-T plate lines. Read More ![]()
Athenahealth to guarantee stimulus cash for docs
The web-based EMR provider guarantees that physicians using its electronic medical records service will get their share of the “meaningful use” provision in the American Recovery and Reinvestment Act.
Read More ![]()
Serica Technologies is adding on to the executive suite
The Medford, Mass.-based tissue repair firm adds plastic surgery and advance manufacturing VPs to its management roster, hiring John Gross and Ramesh Rao, respectively. Read More ![]()
Medtronic loses $30 million insulin pump suit against Nova Biomedical
The Minneapolis-based devices giant had sued to stop Waltham, Mass.-based Nova from selling wireless insulin meters and test strips compatible with Medtronic MiniMed’s insulin pumps. Read More ![]()
Thermo Fisher CEO Dekkers to helm Bayer
The Waltham, Mass.-based lab instruments maker’s chief is tapped to lead the German pharma giant; Thermo Fisher promotes COO Marc Casper to the corner office. Read More ![]()
![]()
Jobs
The MassDevice Career Connector: Open jobs in the medical device industry
Our jobs board has hundreds of open positions at medical device makers around the country. Read More ![]()
![]()
Features
MassDevice Q&A: Richard Schumacher
The Pressure BioSciences founder, president and CEO on how it took seven college majors for him to find his niche, making it through the recession and why his firm’s pressure cycling technology could revolutionize scientific research. Read More ![]()
![]()
Blogs
MassDevice blog: Device makers scurry to derail Baucus tax proposal
The medical device industry is trying by hook or crook to avoid a $4 billion a year tax proposed by Sen. Max Baucus as part of the healthcare reform effort. Read More ![]()
Weekly Wireless Roundup: The real top medical iPhone apps (money-making edition)
Apple’s list of the top 200 highest-grossing applications in its iTunes App Store; healthcare’s wireless sensor opportunity; Corventis completes clinical trial enrollment; and mobile robotics improve patient interactions. Read More ![]()
MedGadget’s MedTech Monday: Is Bloodbot the future of phlebotomy?
The Dept. of Mechanical Engineering at Imperial College London is developing a robot to take blood; Hansen Medical’s Sensei X robotic catheter system; Tulane University’s umbilical cord-cutting SafeSnip tool; and an implanted tooth restores vision in a patient with scarred corneas. Read More ![]()
MassDevice blog: CDC researchers mix avian, Swine Flu
Centers for Disease Control researchers are hedging their bets against the H1N1 influenza virus evolving into a more virulent and deadly strain. Read More ![]()
Compliance Corner: Here we go again
It’s time for another round of reform mania in Washington. But will legislators find real solutions to the problems facing healthcare, or will it be business-as-usual inside the Beltway? Read More ![]()
Dollars & Spence: Wireless connectivity and medical devices
Integrating wireless technology into medical devices presents the industry with both a carrot and a stick: Stimulus cash (carrot) versus increasing demand for connective devices from hospitals (stick). Read More ![]()
![]()
Upcoming Events
Sept. 22-24, Boston — EmTech@MIT
The annual EmTech@MIT conference gives you access to the most innovative labs, companies, and entrepreneurs from around the world and shows you where to lead your organization in order to stay ahead of the curve. The conference showcases the most important emerging technologies and explains their impact. It’s packed with groundbreaking demos, thought-provoking keynotes, interactive breakout sessions and opportunities for exclusive networking with our senior-level audience.
Sept. 23, Boston — Massachusetts Life Sciences Center Board Meeting
The next monthly meeting of the MLSC Board will include consideration of a capital investment at the University of Massachusetts Medical School. 1 Ashburton Place, 21st floor conference room.
Sept. 24, Devens, Mass. — WPI Venture Forum — Networking event for entrepreneurs: Bionostics
The WPI Venture Forum, an organization devoted to developing technology-based entrepreneurship, is pleased to announce the continuation of the WPI Venture Forum Networking Series. These networking-only events, held throughout the year, are designed to help entrepreneurs make connections in the community, reestablish old links, and get an inside look at innovative Massachusetts corporations.
The first event of the 2009-2010 season will be held on Thursday, September 24, 5:30-7:30 pm at Bionostics, an FDA registered, ISO9001 designer and cGMP manufacturer of liquids for critical care, point of care, and diagnostic test systems. The evening will include tours of the Bionostics facility in Devens and networking with entrepreneurs, investors, and industry experts.
Sept. 29, Boston — From the Reebok Pump to the insulin pump
MassDevice.com and Boston University’s College of Engineering present a discussion on turning engineering challenges into product innovation with Continuum, a global innovation and design consultancy, starting at 6 p.m. in the Photonics Building, 8 St. Mary’s Street, Rm. 211, Boston. Register at brian@massdevice.com.
Oct. 2, Boston — MassMEDIC’s Annual Investors Conference
“New England is a hotbed of cutting-edge research in the medtech industry. MassMEDIC is proud to offer an opportunity for regional promising early stage companies to showcase their technologies before an audience of potential investors and strategic partners. The Investors Conference has been a seminal event for medtech start-ups, more than 250 companies have participated in the Conference since 1999 — many making their debut before investors.”
Oct. 3, Cambridge, Mass. — Partners in Health Annual Symposium
Speakers this year will include New York Times columnist and Pulitzer Prize-winning journalist Nicholas Kristof, PIH co-founders Paul Farmer and Ophelia Dahl and speakers from PIH partner projects from around the world.
Oct. 7, Waltham, Mass. — Medical Development Group Forum – Merging Mindsets: Device-Biotech Combination Products
Three experienced professionals in this area with different perspectives will make presentations that include a survey of drug-device combinations now being marketed and in the pipeline. Unmet medical needs that could potentially be treated with novel drug-device combinations. The kinds of critical issues that drug-device development teams must understand and address. Case history of successful drug-device product development.
Oct. 27-28, Waltham, Mass. — Medical Product Outsourcing Symposium East
The practice of outsourcing continues to present challenges to medical devices manufacturers. This event is a high-level educational and networking program, designed to offer relevant information for solving business-critical issues in an environment that fosters interaction and industry development.
Oct. 27-28, Boston — CIMIT Innovation Congress 2009
“Accelerating Healthcare Solutions Through Technology: The CIMIT Innovation Congress” is the premier event catalyzing innovations in medical device technology. It’s the only place where academia, government, industry, and the military convene to learn and network with national leaders who are shaping tomorrow’s healthcare solutions.
Oct. 26-27, Waltham, Mass. — New England – Israel Life Sciences Summit
The New England-Israel Life Sciences Summit at the Brandeis International Business School will feature a select number of cutting-edge Israeli life sciences companies. The program will include networking opportunities and company presentations, followed by one-on-one meetings with potential research and collaboration partners, and select prospective investors.
Keynote Speaker: Israel Makov, former CEO of Teva Pharmaceuticals and Chairman Given Imaging, and Guest Speaker: Dr. Paul Bleicher, Chief Medical Officer, Insight Health Technologies and co-founder Phase Forward: and The Honorable Governor of Massachusetts, Deval Patrick is invited and is expected to address the attendees. Pre-registration is required; visit www.neibc.org or contact info@neibc.org or 617-610 0077 for more information.
Want to list your event on our weekly newsletter? Contact us at editor@massdevice.com.
![]()
If you would like to subscribe to this newsletter, please visit our member registration page. ![]()
© 2009 Massachusetts Medical Devices Journal LLC and its licensors. All rights reserved. The material on this site may not be reproduced, distributed, transmitted, cached or otherwise used, except with the prior written permission of MMDJ.




Leave a Reply
You must be logged in to post a comment.