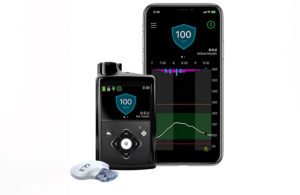
Last month, Medtronic received an FDA warning letter highlighting inadequacies in specific medical device quality system requirements at its diabetes business’ Northridge, California, facility. Today at the 40th Annual J.P. Morgan Healthcare Conference, CEO Geoff Martha said there is now some uncertainty over the timing of approval for next-generation platforms, including the MiniMed 780G insulin pump and Guardian 4 continuous glucose monitoring (CGM) sensor.
Get the full story at our sister site, Drug Delivery Business News.

