
Just a few days into his tenure as CEO of Medtronic (NYSE:MDT), Omar Ishrak was confronted with a major crisis: The Spine Journal devoted its entire June issue to to exposing problems with growth proteins, including a repudiation of some of the research surrounding Infuse.
The ensuing clamor, which included federal investigations into allegations that Medtronic’s paid consultants may have concealed the risks of Infuse, prompted 1 of Ishrak’s first public statements as the new face of Medtronic.
“For several years Medtronic has been leading the industry in reforms designed to eliminate or mitigate conflicts of interest. We will continue to investigate questions surrounding researchers’ potential conflicts of interest, refine our policies as warranted, and strive to lead the industry in ethical and transparent business practices,” he said, and soon put his money where his mouth was.
Sign up to get our free newsletters delivered right to your inbox
Medtronic sponsored a $2.5 million review of the Infuse data by Yale University. Led by rock-star scientist Dr. Harlan Krumholz, the Yale probe will get access to the entire data set from Medtronic’s trials of the Infuse compound, called recombinant bone morphogenetic protein-2 (rhBMP-2).
When MassDevice caught up with 55-year-old Ishrak after his presentation at the J.P. Morgan health care conference in San Francisco yesterday, he seemed surprisingly unfazed by the Infuse kerfluffle.
“This didn’t require me to go and sleep on it overnight, or anything like that. It was just how I think, and how the company thinks,” he told us. “It was something that obviously you don’t want to have to face, but having to deal with it, to me, was in many ways pretty straightforward.”
That’s as good a word as any to describe the 55-year-old Ishrak, who was tapped to replace the retiring William Hawkins last summer. During a wide-ranging chat spanning his handling of Infuse, his 3-point plan to re-invigorate Medtronic, the divestiture of Physio-Control and how he screens potential acquisitions, he took the same even, no-nonsense approach to our questions.
MassDevice: You said today that Medtronic has about $2 billion a year to spend on acquisitions and will look mostly outside the U.S. for them. What regions or treatment areas are you targeting?
OI: I think there’s a number of questions that have to be answered as these opportunities arise. We’re broad enough, with enough options, that I really don’t want to rule anything out. So I look at and encourage our business leaders and the team in general to come up with ideas. I don’t really want to restrict that too much, but I do want to have certain screens.
Some of the screens are, whatever it is, it must a compelling value proposition, with a business team which has a clear plan. At the same time, we’ve got to be responsible about any forms of earnings dilution. It’s not that we can’t have any deals which have earnings dilution, but we’ve got to make sure that we understand that’s the same as another program somewhere, as an internal investment, and we’ve got to make the tradeoff so that overall our productivity doesn’t get hampered by doing any kind acquisition.
And then finally from a market space perspective, we’ve got to be in growth markets, so we’ve got to make sure the market is one that we can justify growth in that market from a long-term perspective. We’ve got to have a clear plan through which we can be a leader in that market. We’ve got to be a leader in the market spaces we’re in; if we’re not, we’ve got to plan to get there.
Finally, there has to be enough value added from Medtronic.
So there are a number of screens that I look at and, independent of whether they’re in the U.S. or outside the U.S. or in this disease or that disease, it has to pass all of those. So that will immediately start to favor some of the shorter-term deals, which have to be what we call bolt-on or tuck-in acquisitions, because business teams are there and you can kind of pass these on fairly easily – the value propositions are clear, they’re probably accretive much more quickly or your business team can make the tradeoffs due to covering the dilution, if any.
So there’ll be some of those and the bigger ones we’ll look at, but it has to also meet certain requirements. That’s how I look at acquisitions.
MassDevice: You gave a clear picture this morning of your strategy to re-focus Medtronic: Improve execution, optimize innovation and accelerate globalization. Based on those criteria, how would you grade the company as you found it, and how would you evaluate your tenure so far?
OI: I think, clearly, those are areas for improvement, because that’s why they were identified, so the incoming grade is not where I want it to be.
As far as my grade is concerned, I think it’s more appropriate to think in terms of what kind of progress we’ve made and how much of a challenge it’s been to implement some of these thoughts.
So let’s take them one by one. I think what’s been, overall, very pleasantly surprising – I mentioned the clinical and technical depth the business has, but in addition, data availability has not been a problem. So looking at where the business stands today and how we measure people, we take business action as a result of both our planning process and our current status. From a financial perspective, I think that’s very good.
Creating a structure through which people can execute – people have received it well and, to be realistic, I’m not sure I could have done it any quicker. I’m still in the process of rolling it out, these structures by definition have a time rhythm to them, and until you’re through a couple of quarters you don’t really know how well they’ve been accepted. So although I feel impatient about it, I think to be realistic it’s going out about as quickly as what we’d expect.
I think in terms of optimizing innovation and economic value, I’ve been pleasantly surprised at the core capability of health care economics that the company has. The ability of the company to grasp the concept of customer economics has been pretty good. But again, changing messaging, gathering evidence, is retooling and in the middle of doing that again it just takes a certain amount of time to do, but it is something that we are making progress with.
With globalization, I think there perhaps the biggest culture change needs to happen. There’s a lot of change in behavior that has to be seen, in terms of people traveling to other countries. I’m seeing a lot of it – when I talk to the regional teams, they tell me there’s more interest from the business units than they’ve ever had, that people are going [overseas] on a regular basis. So there’s a lot of excitement among engineers as to how to develop products for those regions, there’s a lot of activity, but I think that perhaps is the biggest single challenge – how to change the overall culture – and that’s the one I’m working most closely on. That’s the one that will require a lot of work.
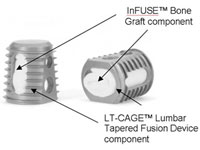
MassDevice: I have to ask about the Infuse controversy, which erupted shortly after you stepped in. What was it like trying to get your feet under you at a new job while trying to navigate that crisis?
OI: It was pretty straightforward. What I said there was something I would have said any time any situation like that was presented, so I didn’t have too much thinking to do.
When you are faced with a situation where the data is confused, you’ve got to get data. Where people are challenging our integrity and our commitment to safety, we’ve got to be absolutely clear and certain, internally and externally, that that’s not a compromise. So coming out with those 3 things, essentially, and sticking to it was all I did. I think that helped, in retrospect, to stabilize the situation.
The Yale study is getting the data piece in a transparent way, making clear that integrity and patient safety are basic principles and being crystal-clear about it. And being completely consistent internally and externally – and not only saying it, but doing it – was critical.
But that was a gut reaction. This didn’t require me to go and sleep on it overnight, or anything like that. It was just how I think, and how the company thinks, so there was no push-back and everyone agreed and we moved forward. My colleagues, such as [chief scientific, clinical and regulatory officer Dr.] Rick Kuntz and others – [restorative therapies president] Chris O’Connell – we talked about it, because my thought originally was, how do I get independent data? And I wasn’t familiar enough with the market to see how to actually do it. But Rick, fortunately, is an expert in this area and he’s very critical to the industry and in that community, and he came up with the Yale idea. I was completely supportive, so as a team we decided to put that in place and I think that was the right thing to do.
So again, it was something that obviously you don’t want to have to face, but having to deal with it, to me, was in many ways pretty straightforward.
MassDevice: The company was looking to shed Physio-Control long before you came along as CEO – what was is that enabled you to get a deal done so soon after taking over the corner office?
OI: Well, they were already working on it. What I made clear is what are the criteria and then the company, the team had already been thinking about it. Clarifying some of the key screens with which we look at businesses made the decision-making pretty direct.
MassDevice: Can you use those screens in reverse when considering divestitures?
OI: Absolutely, it’s the same questions: Are we in a growth market? Can we win? And is Medtronic adding enough value, or is the business better off on its own?
We ask those questions internally on a regular basis and we ask those questions externally on a regular basis. If you look at Physio-Control, is it a growth market? Yeah, probably, it’s in the ER segment, the need for defibrillators is pretty high, so it’s a growth market. Can we win in it? Potentially, but it’ll require a lot of investment. We’re still the number 1 player, so we are actually in a good position to win it. Can Medtronic add value? That’s where we got stuck, because there was no real synergy with anything else.
We could have stayed in that space if we decided that this is the platform to build a business around, the ER or something like that, but we weren’t prepared to do that and so it didn’t really make any sense. To me at least, the divestiture decision was confirmed around the added value of Medtronic. We could argue some, but there weren’t really any real technology synergies, there weren’t any customer synergies, there weren’t any real manufacturing synergies. And even from a distribution perspective, there wasn’t a whole lot, so unless we created a separate platform there really was not too much there.
MassDevice: You’re looking to shave another $1.2 to $1.3 billion from your budget, adding to the 25% reduction that’s just coming to a close now. Can you give us some detail on how you’ll reach that goal?
OI: We’re just beginning that process, so I cannot really give you too much detail, but I’ll tell you some simple philosophies. First, I look at cost reduction as an ongoing exercise. So guess what? In the next 5 years, there’ll be another 25% target, so this is not going to go away.
One of the main messages here is, we’ve got to look at continuous cost reduction. And I look at product cost reduction in a continuous, 3-phase cycle: First, you optimize your sourcing, your volume and your sourcing, and you go to low-cost countries and optimize your sourcing. After a while, that wears out, so the next thing you do is look at a redesign of specific components that can take into account current availability of components. Then the 3rd thing you do is look at transformational design, major changes in architecture, which enables you to reduce cost and improve performance at the same time.
The computer industry does this all the time; we’ve got a lot of electronics in our products, so changing architecture to reduce costs and improve is always something that we’ve also got to do. It takes little longer, but you’ve got to be doing it, and then you start all over again. Once you introduce new architecture, you go back to sourcing, because you’ve got a whole bunch of new components. This 3-phase cycle is something I want all the business teams to embrace and put into their working methodology.
The methods we’ll use to reduce costs cover some or all of these; in addition, there are other opportunities. Many of the businesses have been relatively recent acquisitions that haven’t been fully integrated, so there are areas for factory consolidation, there are areas of manufacturing and operational efficiency – putting lean in areas where we don’t have it. From a short-term perspective, this low-cost country [idea] is a pretty big opportunity, because we’re well below where I think our entitlement should be.
MassDevice: There’s a lot of uncertainty in the U.S. over the regulatory climate. What’s your take on the environment today and the moves the FDA is taking to reform the 510(k) protocol?
OI: We really want to work with the regulatory authorities to streamline things in a way that we can get good therapies out to market in a more efficient fashion. I think the regulatory authorities are actually very open to that dialogue, in terms of sharing data and in terms of basically doing the right things for patients.
I want to make it clear that we want to partner with them. Patient safety is as much our concern as it is theirs, and we will ensure that. Speed to market is also something that’s valuable both to us and to them, because we are providing therapies that are useful to patients in this country. So clarifying that we have a partnership, rather than an adversarial relationship, I think is very important.
Now what are some of the more concrete things that we’re doing? One of the things that we’re exploring is a much more rigorous post-market surveillance method, which is high on the FDA’s list. We’ve got real expertise around clinical trials and around post-market surveillance and we’re willing to invest, we will invest in this kind of a network, which is beyond a registry where we actually gather clinical data and take action. We’re committed to taking action based on that data and we’ll work with the FDA on that.
It’s something that we’ll do anyway, because it’s something that I think we need to do to be proactive about patient safety. Through that, we can work with the FDA to speed up some approvals, I think that’ll help us out.
Regarding 510(k) and so on, I mean look, we’re primarily a [pre-market approval] company, so that’s really where our focus is and we excel at that.
MassDevice: Stryker made news when it announced a 5% workforce reduction due to the impending medical device tax. You’re more insulated from its effects due to your high proportion of international sales, but I’m still interested in how will Medtronic look to handle the tax.
OI: We’ve got to cover it in some way and, realistically, it’s going to happen. I think to some degree we’ve got to share it with our customers, but we’re going to be pragmatic about it. It’s something that we’ve got manage through.
There’s pressures everywhere, in all businesses, in a variety of ways. There’s pricing pressure for a variety of reasons – that’s why we do our cost reduction and be proactive about our cost reduction and look for efficiencies. This is just 1 additional pressure that we’ve got to manage through.
MassDevice: How involved is Medtronic in lobbying efforts to repeal the tax?
OI: I think, look, we’re part of AdvaMed and we’re working with them and I think that’s about it.

