Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else this weekend, make sure you’re still in the know with MassDevice +7.
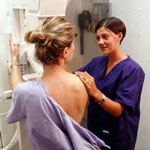
7. Mixed messages on the merits of mammograms
New research on mammogram usage suggests that frequent screening can increase the chances of breast cancer detection, but nearly 1 out of every 3 caught tumors results in unnecessary treatment.
![]()
6. FDA’s medtech review Plan of Action cuts review times, lowers application backlog
The FDA touts the success of its 36-point "Plan of Action" for streamlining the medical device premarket review process, noting improvements in predictability in transparency for the 1st time in nearly a decade.
![]()
5. Baxter’s in the hunt for $4B Gambro buyout
Baxter International is said to be in the hunt for a $4 billion acquisition of Swedish medical device company Gambro.
![]()
4. Smith & Nephew’s $782M Healthpoint buyout raises eyebrows
Smith & Nephew announces a $782 million acquisition for wound care products maker Healthpoint Biotherapeutics, a move that generated mixed reactions for analysts.
![]()
3. Stryker can’t dodge Trident hip lawsuit
A federal judge denies Stryker’s move to quash a product liability lawsuit filed over its recalled Trident metal-on-metal hip implant.
![]()
2. St. Jude shares hit new low following FDA report
Medtech titan St. Jude Medical slides 7% after the FDA releases a report slamming the device maker’s Durata pacemaker leads.
![]()
1. Medtech VC spending in Q3 2012 hits lowest level since 2004
Venture capitalists spend more money on lifesciences deals during the 3rd quarter of 2012, but the medical device segment hits hard times as biotech takes the fore, according to a PricewaterhouseCoopers U.S. report.