Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
7. Q&A with patient safety advocate Dr. Robert Hauser
Veteran cardiologist and patient safety advocate Dr. Robert Hauser tells MassDevice.com about his new vision for healthcare surveillance, his long struggle to spur change at the FDA and what FDA regulators can learn from aviation regulation.
![]()
6. J&J’s Cordis snaps up Flexible Stenting Solutions
Johnson & Johnson’s stenting arm, Cordis Corp. furthers its turn away from coronary stents toward the endovascular market with the acquisition of Flexible Stenting Solutions for an undisclosed amount.
![]()
5. FDA warns Stryker on quality systems, recalls, Neptune marketing
Stryker says the FDA sent it a warning letter over problems with its quality system at a plant in Michigan, failing to notify the federal watchdog agency about recalls and for the marketing of its Neptune waste management device.
![]()
4. FDA warns on arrhythmia risk with ‘Z-Packs’
The FDA warns of the risk of potentially lethal arrhythmias in certain patients who take the antibiotic drug azithromycin, commonly known as “Z-Packs.”
![]()
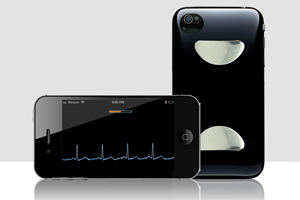
3. FDA clears over-the-counter iPhone ECG
Utah medical device startup Cardiac Designs wins over-the-counter indication for its iPhone-based ECG monitor.
![]()
2. Boston Scientific apologizes for embargo breach that got Watchman data pulled from ACC
American College of Cardiology conference organizers cancel the highly anticipated presentation of preliminary results from Boston Scientific’s Prevail study of its Watchman device after the company accidentally leaks the data ahead of time.
![]()
1. The 6 lines of code that could bring down a hospital
The software flaw that allowed a duo of cybersecurity researchers to bring down a Philips XPER hospital management system with 6 lines of code is still a problem in current XPER machines, Philips tells MassDevice.com.