Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
7. St. Jude’s Durata defibrillator ‘immune’ to Riata flaws?
St. Jude Medical’s Durata implantable cardiac defibrillator leads should be “relatively immune” to the problems that led to a recall for the medical device company’s Riata ICD leads, according to a cardiologist who helped bring the Riata problems to light.
![]()
6. Echo Therapeutics sends pre-submission package to FDA
Echo Therapeutics initiates discussions with the FDA on the regulatory pathway for its Symphony non-invasive, wireless continuous glucose monitor.
![]()
5. Covidien’s Mallinckrodt pharma spinout slated to close this month
Covidien’s spinout of its Mallinckrodt pharmaceuticals business is slated to close later this month.
![]()
4. Medical device tax: Consultant calls offshoring concerns ‘horse hockey’
Concerns that the medical device tax will push companies to lay off workers and move operations overseas are ‘horse hockey,’ according to consultant Mark Bonifacio.
![]()
3. CareFusion may be weighing 2 very different deals

CareFusion seems to be a key potential acquirer for at least 2 medical device businesses known to be on the block – ICU Medical and Smiths Group’s Smiths Medical.
Read more
![]()
2. Real-world results cast doubt on renal denervation for high blood pressure
Study results revealed in Europe this week cast doubt on the efficacy of renal denervation for treating high blood pressure.
![]()
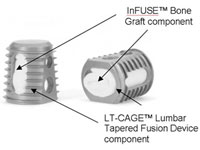
1. Yale study of Medtronic’s Infuse shows it’s equivalent to bone graft, but studies were biased
A pair of independent analyses of patient-level data commissioned by a Yale University open data program show that Medtronic’s Infuse bone-growth putty is equivalent to bone grafts, with 1 concluding that early Infuse research was biased.