Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
3. FDA expands indication for Medtronic’s StealthStation nav suite
Medtronic said surgeons can now use its StealthStation navigation tool in cranial and ENT surgeries in children and adults, after the FDA expanded the indication for the device.
StealthStation is designed to establish an electromagnetic field that can be used to pinpoint the location of instruments and markers set to locate anatomical structures, Medtronic said. Read more
![]()
![]()
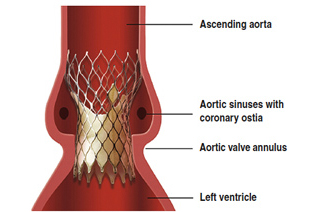
2. TAVI: U.K. study finds 60% survival rate at 3 years
The survival rate for high-risk patients with transcatheter aortic valve implants surpassed 61% at 3 years and drops to 45.5% at 5 years, according to a new analysis of data from the U.K. Transcatheter Aortic Valve Implantation Registry.
The results were published in the latest issue of JACC: Cardiovascular Interventions, featuring the work of U.K. researchers from the Royal Brompton Hospital, Queen Elizabeth Hospital and University College London, among others. Read more
![]()
1. Bayer: 5-year results confirm Essure’s safety and effectiveness
Bayer Healthcare cited a study published in the Journal of Minimally Invasive Gynecology with 5-year results for its Essure female sterilization procedure, which has become a lightning rod for patient activists since the FDA approved it 13 years ago.
Bayer said the study, a prospective 5-year follow-up of the initial 364-patient cohort from its Phase III trial for Essure, showed no pregnancies after 5 years and that the Essure inserts were “generally well tolerated.” The Phase III trial enrolled fertile women ages 21 to 40 in monogamous relationships who were willing to use a temporary contraceptive for the 1st 3 months after Essure implantation, Bayer said. Read more