Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
3. FDA panel issues mixed vote on EnteroMedics weight-loss implant
EnteroMedics got a mixed bag of support from a panel of FDA advisors yesterday, with most panelists voting that the company’s weight-loss implant is safe but raising concerns about the effectiveness of the device.
The 9-member panel voted 8-1 that EnteroMedics’ Maestro nerve-stimulation implant is safe and 6-2 with 1 abstention that the overall benefits outweigh the risks, but the panel split 4-5 on whether the device is effective. The FDA isn’t bound by the recommendations of its advisory panels, but the federal watchdog agency usually sides with them. Read more
![]()
![]()
2. Device makers fight pricing war with new study
Medical device makers have long faced intense pricing pressure from customers and scrutiny from policy-makers, but an ongoing industry study suggests that medtech has for years been a minor factor in the growing cost of U.S. care.
Researchers for industry lobbying group AdvaMed released the 2nd edition of their pricing study, reporting that medtech represents a reliably small portion of total U.S. healthcare spending. Devices spending has grown from 5.3% to 5.9% of total national healthcare spending over a 23-year period spanning from 1989 to 2011, and most of that increased occurred before 1992, according to the researchers. Furthermore, their analysis found that medical devices have grown far more slowly than the Medical Consumer Price Index. Read more
![]()
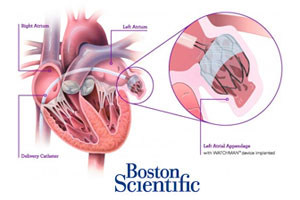
1. FDA wants another panel hearing on Boston Scientific’s Watchman anti-stroke device
Boston Scientific CFO Dan Brennan said that the FDA wants to hold another advisory panel hearing on its Watchman anti-stroke device and pushed the approval timeline for the Watchman heart implant into next year. Brennan, speaking at the Wells Fargo Healthcare Conference, said Boston Scientific is now hoping for an approval during the 1st half of 2015.
The as-yet-unscheduled panel would be the 3rd advisory committee hearing over Boston Scientific’s pre-market approval application for the Watchman device and comes despite positive votes from the 2 prior meetings. Read more